Experimental studies on reduction desulfurization of FGD gypsum from iron ore sintering process using CO/H2
-
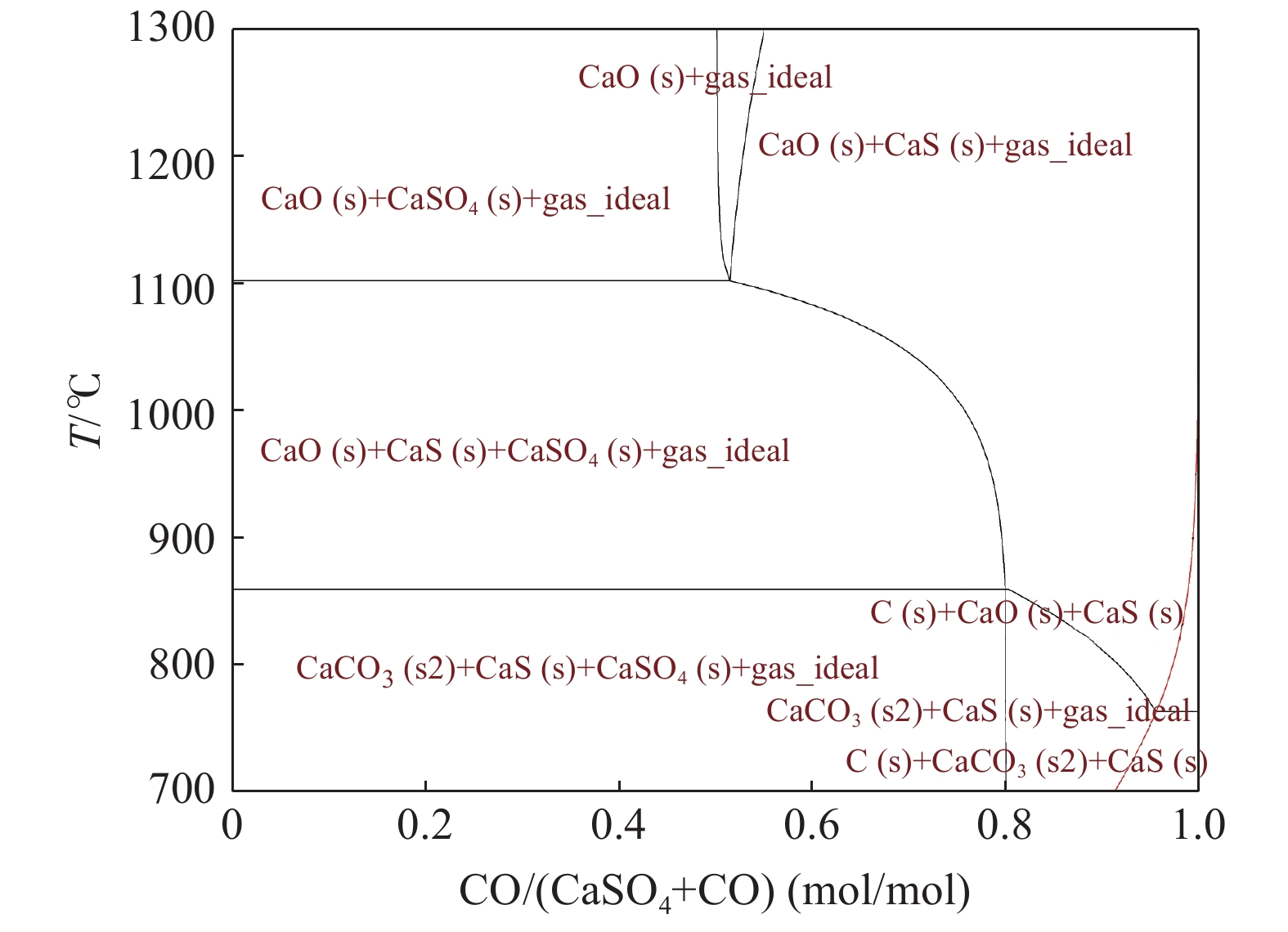
摘要: 通过热力学软件Factsage7.3对CO/H2还原CaSO4进行热力学平衡计算。计算得到CO/H2还原CaSO4生成CaO和SO2的最佳理论条件为:反应温度1100 ℃以上、摩尔比n(CO)∶n(CaSO4)=n(H2)∶n(CaSO4)=1,过高或过低的还原剂摩尔比均不利于CaSO4的还原脱硫,同时温度越高越有利于CaSO4的CO / H2还原脱硫。根据Factsage7.3计算结果,以N2作为混合气体,在高温炉中进行了15 g烧结烟气脱硫石膏的CO/H2还原脱硫试验,考察CO/H2浓度、反应温度、反应时间、还原气体流量对脱硫石膏CO/H2还原脱硫的影响。结果表明:有利于脱硫石膏CO还原脱硫的最佳条件为CO浓度5%、反应温度1050 ℃、反应时间30 min、气体流量5 L/min;有利于脱硫石膏H2还原脱硫的最佳条件为H2浓度8%、反应温度1050 ℃、反应时间30 min、气体流量为5 L/min,且CO分解还原脱硫效果较H2还原脱硫的效果要好。试验发现在900 ℃以上时脱硫石膏有烧结现象发生,且随温度升高烧结程度更重,影响了CO/H2的还原脱硫效率。Abstract: The thermodynamic equilibrium of CO/H2 reduction of CaSO4 was calculated through software FactSage 7.3. The optimum theoretical conditions for generation of CaO and SO2 by CO/H2 reduction of CaSO4 were obtained at more than 1100 ℃ of reaction temperature and mole ratio of n(CO)∶n(CaSO4)=1 or n(H2)∶n(CaSO4)=1. The reduction agent with a too high or too low mole ratio (n(CO)∶n(CaSO4) or n(H2)∶n(CaSO4)) is not beneficial to the reduction desulfurization of CaSO4. A higher temperature is favorable for the CO/H2 reduction desulfurization of CaSO4. According to the thermodynamic calculation results, experimental studies on reduction desulfurization of FGD gypsum from iron ore sintering process were carried out using CO/H2+N2 mixture gas in a furnace, and the influences of CO/H2 concentration, reaction temperature, reaction time and gas flow rate on the reduction desulfurization of FGD gypsum by CO/H2 were studied. The results show that the beneficial conditions for CO reduction desulfurization of FGD gypsum are CO concentration of 5%, temperature of 1050 ℃ for 30 min and gas flow rate of 5 L/min. The beneficial conditions for H2 reduction desulfurization are H2 concentration of 8%, temperature of 1050 ℃ for 30 min and gas flow rate of 5 L/min. The efficiency of reduction desulfurization by CO is better than that by H2. During the reduction process, FGD gypsum sinters above 900 ℃ and it makes a notable impact on the reduction desulfurization of the FGD gypsum.
-
Key words:
- sintering /
- flue gas /
- desulfurization gypsum /
- reduction desulfurization /
- CO /
- H2 /
- desulfurization ratio
-
表 1 烧结烟气脱硫石膏化学成分
Table 1. Compositions of FGD gypsum
% CaO SiO2 MgO Al2O3 TFe P (SO3) 全S 38.40 1.71 0.21 0.25 0.23 ≤0.003 (51.75) 20.70 -
[1] Meng Lingjia, Ji Zhonghai, Chen Jin. Advance of the thermal decomposition of industrial by-product gypsum[J]. Chemical Industry and Engineering Progress, 2017,36(2):626−633. (孟令佳, 吉忠海, 陈津. 工业副产石膏热分解脱硫的研究进展[J]. 化工进展, 2017,36(2):626−633. [2] Han Lingcui, Wang Yongchang. Development of chemical gypsum industry depends on the integration of energy saving and emission reduction with comprehensive utilization[J]. Phosphate & Compound Fertilizer, 2010,25(6):11−13, 21. (韩灵翠, 王永昌. 节能减排与综合利用并重是化学石膏产业发展方向[J]. 磷肥与复肥, 2010,25(6):11−13, 21. doi: 10.3969/j.issn.1007-6220.2010.06.003 [3] Hu Zhenyu, Wang Jian, Zhang Xian, et al. Comprehensive utilization of phosphgypsum[J]. China Mine Engineering, 2004,33(4):41−44. (胡振玉, 王健, 张先, 等. 磷石膏的综合利用[J]. 中国矿山工程, 2004,33(4):41−44. doi: 10.3969/j.issn.1672-609X.2004.04.012 [4] Richard R. West and Willard J. Sutton Thermography of gypsum[J]. Journal of the American Ceramic Society, 1954,37(5):221−224. doi: 10.1111/j.1151-2916.1954.tb14027.x [5] Miao Zhu, Yang Hairui, Wu Yuxin, et al. Experimental studies on decomposing properties of desulfurization gypsum in a thermogravimetric analyzer and multiatmosphere fluidized beds[J]. Industrial & Engineering Chemistry Research, 2012,51(15):5419−5423. [6] Xiao Haiping, Zhou Junhu, Cao Xinyu, et al. Experiments and model of the decomposition of CaSO4 under CO atmosphere[J]. Journal of Fuel Chemistry and Technology, 2005,33(2):150−154. (肖海平, 周俊虎, 曹欣玉, 等. CaSO4在CO气氛下的平行竞争反应试验与模型研究[J]. 燃料化学学报, 2005,33(2):150−154. doi: 10.3969/j.issn.0253-2409.2005.02.005 [7] Su Hang, Zuo Haibin, Zhao Jun. Desulphurization of gypsum at high temperature[J]. Inorganic Chemicals Industry, 2019,51(7):68−73. (苏航, 左海滨, 赵骏. 石膏在高温下的分解脱硫研究[J]. 无机盐工业, 2019,51(7):68−73. doi: 10.11962/1006-4990.2018-0486 [8] Zheng Shaocong, Ning Ping, Wang Fan, et al. Study on preparation of sulfur dioxide and lime by thermal decomposition of phosphogypsum[J]. Inorganic Chemicals Industry, 2013,45(9):45−47. (郑绍聪, 宁平, 汪帆, 等. 磷石膏热分解制备二氧化硫和氧化钙研究[J]. 无机盐工业, 2013,45(9):45−47. doi: 10.3969/j.issn.1006-4990.2013.09.015 [9] Han Xiangyu, Chen Haokan, Li Baoqing. Thermogravimetric study on the reductive decomposition of calcium sulfate with pure H2 at high temperature[J]. Coal Conversion, 2000,23(2):72−75. (韩翔宇, 陈皓侃, 李保庆. CaSO4氢气下还原分解的热重研究[J]. 煤炭转化, 2000,23(2):72−75. doi: 10.3969/j.issn.1004-4248.2000.02.014 [10] Chen Sheng, Liu Shaowen. Thermodynamic study on reductive decomposition of calcium sulfate with hydrogen[J]. Journal of Chemical Industry & Engineering, 2012,33(5):7−11. (陈升, 刘少文. 氢气还原分解硫酸钙的热力学研究[J]. 化学工业与工程技术, 2012,33(5):7−11. [11] Shao Xing, Gong Lei, Cao Yuexi, et al. Study on properties of FGD gypsum from iron sintering process[J]. Zhejiang Metallurgy, 2018,(1):28−32. (邵兴, 巩磊, 曹悦曦, 等. 烧结废气脱硫石膏性能探究[J]. 浙江冶金, 2018,(1):28−32. [12] Deng Bo, Gong Lei. Preliminary experimental study on decomposition and reduction of FGD gypsum from iron sintering process[J]. Zhejiang Metallurgy, 2019,(1):9−13. (邓波, 巩磊. 烧结废气脱硫石膏分解与还原试验初探[J]. 浙江冶金, 2019,(1):9−13. -





 下载:
下载: