New process for utilization of sea sand vanadium-bearing titanomagnetite
-
摘要: 为提高海砂钒钛磁铁矿的综合利用效率,降低处理能耗,提出了基于转底炉直接还原-电炉熔分流程的活化还原处理方法,并采用化学分析方法考察了铁、钒、钛的提取规律,优化了工艺参数,提出了改善电炉熔炼效率的措施。活化还原-电炉熔分方法可有效提高海砂钒钛磁铁矿的还原和熔分效率,并促进铁水的脱硫过程,活化还原温度控制在1150~1200 ℃、电炉熔分温度控制在1450 ℃时效果最佳。活化还原为电炉熔分与吹氧提钒相结合创造了条件,可显著降低熔分电耗、提高钒的提取效率,并简化炼钢流程。新的处理流程下熔分钛渣的活性较高,在体积浓度高于15%的稀硫酸中钛的浸出率可达95%,避免了传统硫酸法提钛过程的废酸排放。
-
关键词:
- 海砂钒钛磁铁矿 /
- 转底炉直接还原-电炉熔分 /
- 活化还原 /
- 回收率
Abstract: In order to improve the comprehensive utilization efficiency of sea sand vanadium titanomagnetite and reduce the treatment energy consumption, an activated reduction treatment method based on the rotary hearth furnace (RF) reduction-electric furnace (EAF) smelting separation process was put forward. The extraction of iron, vanadium and titanium was investigated to optimize the process parameters, and a measure improving the smelting efficiency of EAF was proposed. The smelting efficiency of the sea sand vanadium titanomagnetite as well as the desulfurization limit of the molten iron are improved effectively in the activated reduction-EAF smelting separation flow, in which the optimum effect can be acquired with the activated reduction temperature at 1150~1200 ℃ and the EAF smelting temperature at 1450 ℃, respectively. Moreover, the activated reduction promotes vanadium extraction by oxygen blowing in the EAF smelting process, significantly reducing the power consumption of EAF, improving the extraction efficiency of vanadium and simplifying the steel-making process. Under the new treatment process, the activity of titanium-bearing molten slag is improved, with the titanium leaching rate in dilute sulfuric acid (volume concentration higher than 15%) reaching 95% which avoids the waste acid discharge in the traditional sulfuric acid extraction process. -
表 1 海砂钒钛磁铁矿的化学成分
Table 1. Chemical compositions of sea sand vanadium titanomagnetite
% TFe FeO Fe2O3 TiO2 V2O5 SiO2 CaO MgO Al2O3 K Na Cl 56 21.85 56 10.67 0.76 4.29 0.72 3.18 4.12 <0.1 <0.1 <0.05 表 2 无烟煤粉化学组成
Table 2. Chemical compositions of pulverized anthracite
% 固定碳 挥发分 灰分 S 水分 74.53 5.16 17.86 0.44 2.45 表 3 原料的粒度组成
Table 3. Particle size distribution of raw materials
粒度/目(μm) 含量/% 海砂钒钛磁铁矿 无烟煤粉 >35(500) 0.4 3.78 35~50(500~297) 0.36 4.53 50~100(297~147) 26.45 65. 6 100~200(147~74) 69.07 27.3 <200(74) 1.72 0.78 表 4 含碳复合球团物料配比情况
Table 4. Mass fractions of carbon composite pellets
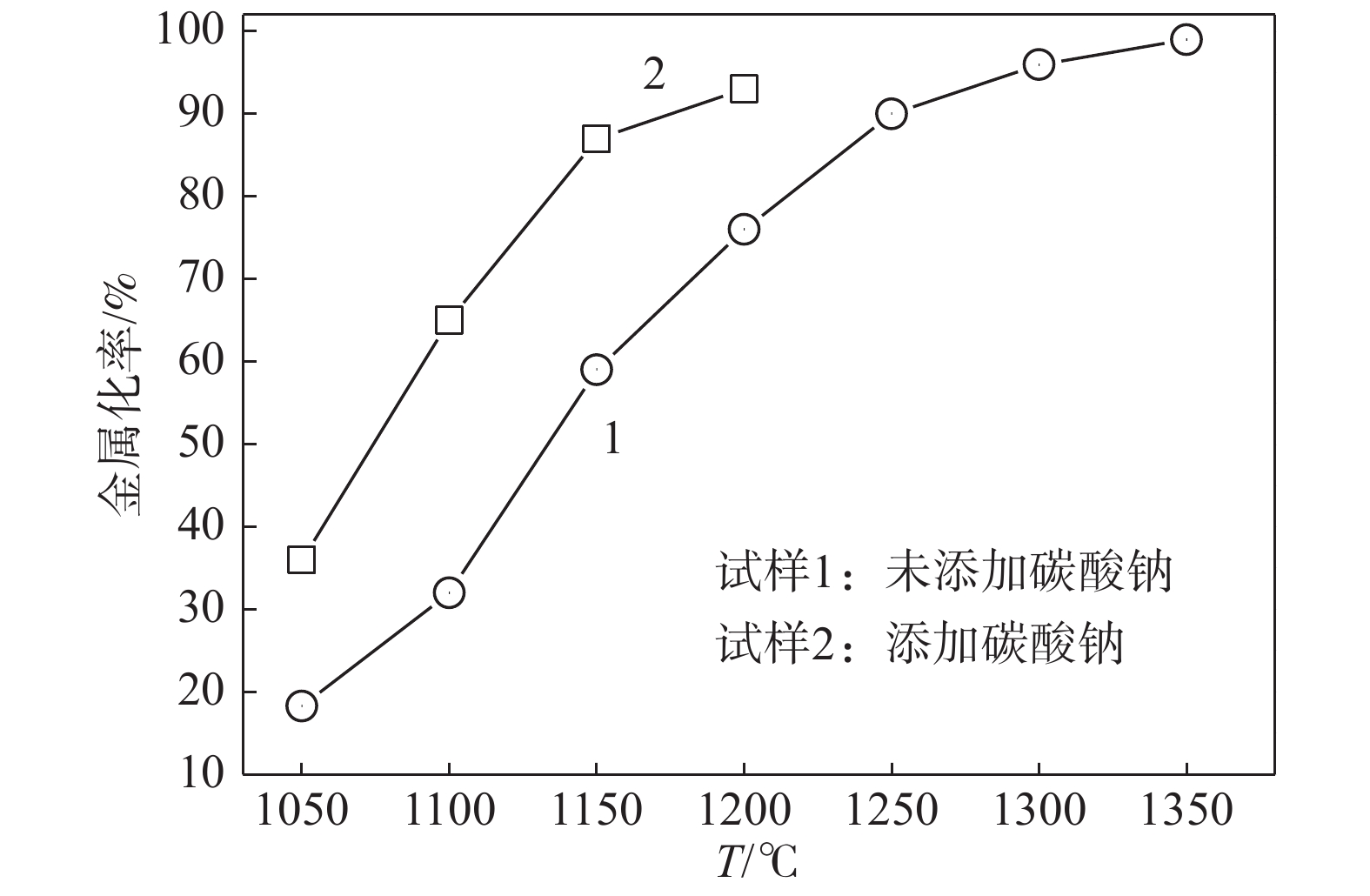
% 试样 碳酸钠 海砂矿 煤粉 1 0 79 21 2 23 61 16 表 5 不同条件下的粒铁成分
Table 5. Chemical compositions of iron nuggets under different conditions
试样 熔分温度/℃ V/% C/% S/% 1 1700 0.32 5.28 0.066 2 1450 0.68 4.87 0.012 -
[1] Ren Qianqian, Hao Suju, Jiang Wufeng, et al. Study of comprehensive utilization on Ti-bearing blast furnace slag[J]. Applied Mechanics and Materials, 2014,488-489:141−144. doi: 10.4028/www.scientific.net/AMM.488-489.141 [2] Liu Mingpei. Analysis of vanadium distribution regularity in Panzhihua vanadium-titanium magnetite mine[J]. Mining Engineering, 2009,7(5):9−12. (刘明培. 浅谈攀枝花钒钛磁铁矿钒的分布规律[J]. 矿业工程, 2009,7(5):9−12. doi: 10.3969/j.issn.1671-8550.2009.05.004 [3] Han Zhibiao, Chang Fuzeng. Material problem and solutions about further development of titanium industry in China[J]. Titanium Industry Progress, 2012,29(1):5−8. (韩志彪, 常福增. 中国钛工业发展的原料问题及对策[J]. 钛工业进展, 2012,29(1):5−8. doi: 10.3969/j.issn.1009-9964.2012.01.002 [4] Zhang Jun, Dai Xiaotian, Yan Dingliu, et al. Carbothermal sodium reduction process of vanadium-bearing titanomagnetite[J]. Iron and Steel, 2016,51(10):6−9. (张俊, 戴晓天, 严定鎏, 等. 钒钛磁铁矿碳热钠化还原工艺[J]. 钢铁, 2016,51(10):6−9. [5] Chen Qianye, Zhang Jun, Cheng Xiangkui, et al. Study on carbothermal sodium reduction process of vanadium-titanium magnetite concentrate[J]. Iron Steel Vanadium Titanium, 2017,38(2):11−15. (陈乾业, 张俊, 程相魁, 等. 钒钛磁铁精矿低温综合利用新工艺[J]. 钢铁钒钛, 2017,38(2):11−15. doi: 10.7513/j.issn.1004-7638.2017.02.002 [6] Zhang Y M, Yi L Y, Wang L N, et al. A novel process for the recovery of iron, titanium, and vanadium from vanadium-bearing titanomagnetite: sodium modification–direct reduction coupled process[J]. International Journal of Minerals, Metallurgy and Materials, 2017,24(5):504−511. doi: 10.1007/s12613-017-1431-4 [7] Zhang Y M, Wang L N, Chen D S, et al. A method for recovery of iron, titanium, and vanadium from vanadium-bearing titanomagnetite[J]. International Journal of Minerals, Metallurgy and Materials, 2018,25(2):131−144. doi: 10.1007/s12613-018-1556-0 [8] Wu X, Chen D S, Wang L N, et al. Study on one-step coal-based direct reduction and melting of vanadium titanium magnetite concentrate[J]. Nonferrous Metals(Extractive Metallurgy), 2019,(2):15−20. [9] Shi L Y, Zhen Y L, Chen D S, et al. Carbothermic reduction of vanadium-titanium magnetite in molten NaOH[J]. ISIJ International, 2018,58(4):627−632. doi: 10.2355/isijinternational.ISIJINT-2017-515 [10] Chen Ronggui, Wang Xuewen, Shou Liting. Study on extraction of vanadium by oxidation followed by sodium[J]. Iron Steel Vanadium Titanium, 1985,6(5):32−35. (陈荣贵, 王学文, 寿煜庭. 先氧化后钠化提钒工艺的研究[J]. 钢铁钒钛, 1985,6(5):32−35. -





 下载:
下载: