Accelerated search for solid lithium-ion conductor materials based on material genome engineering
-
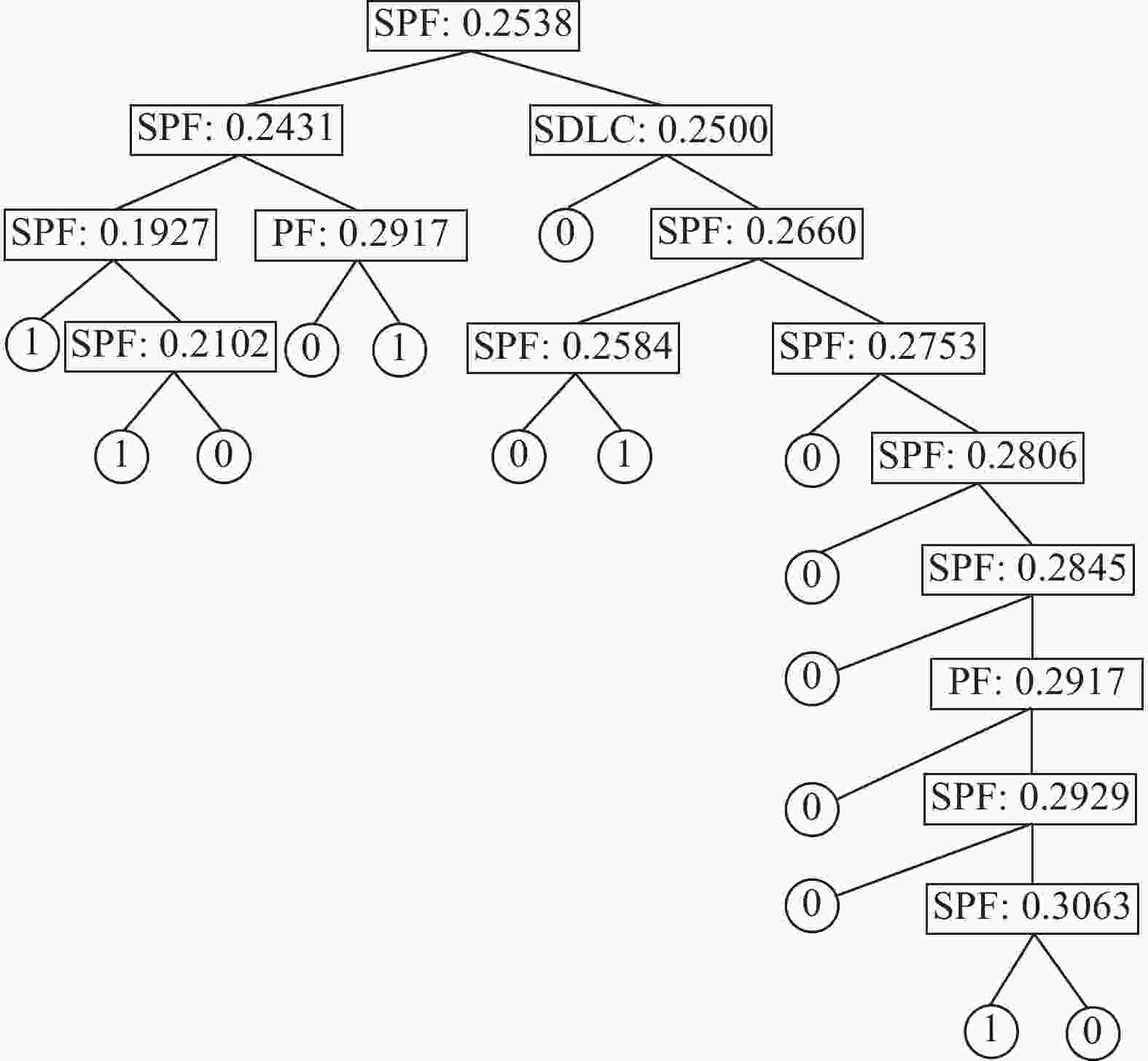
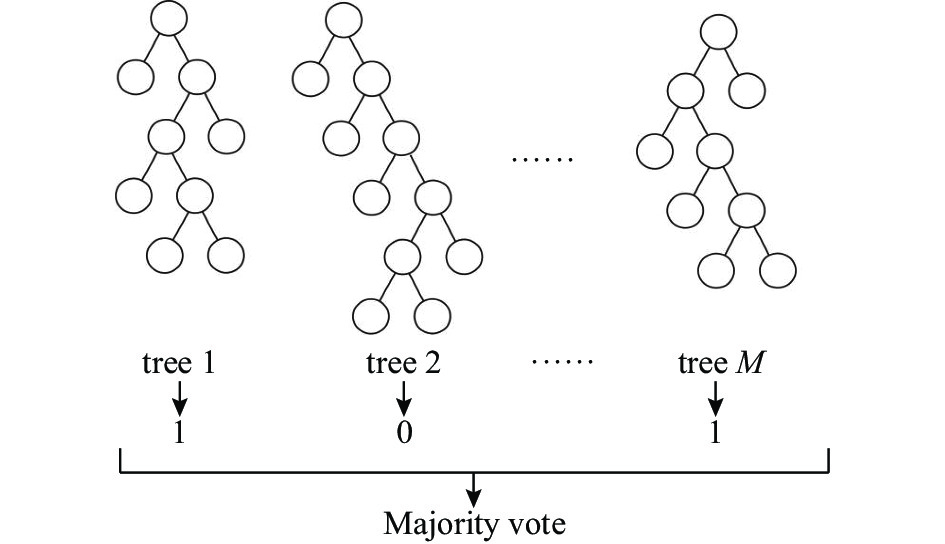
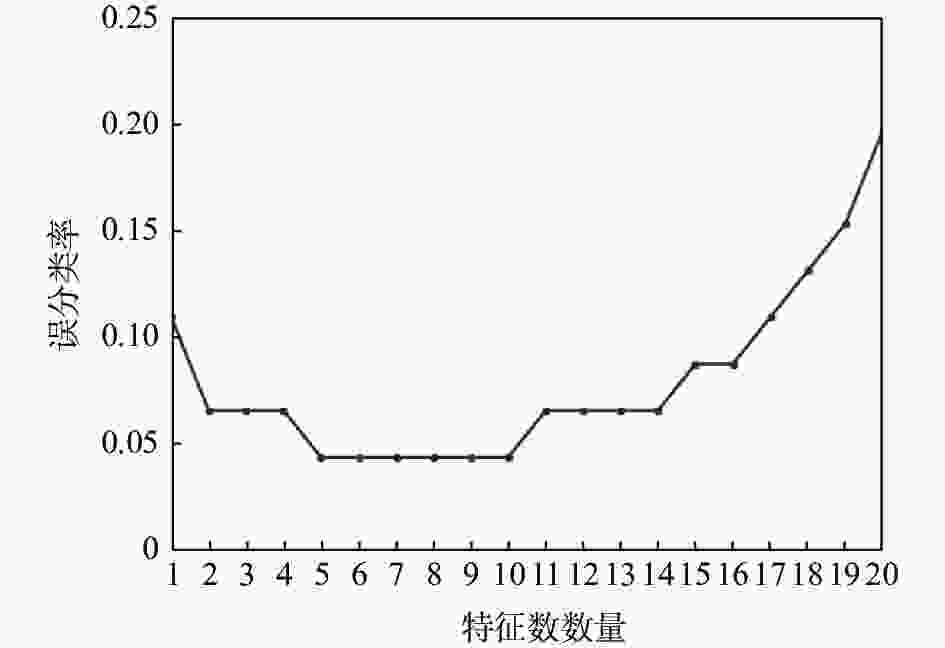
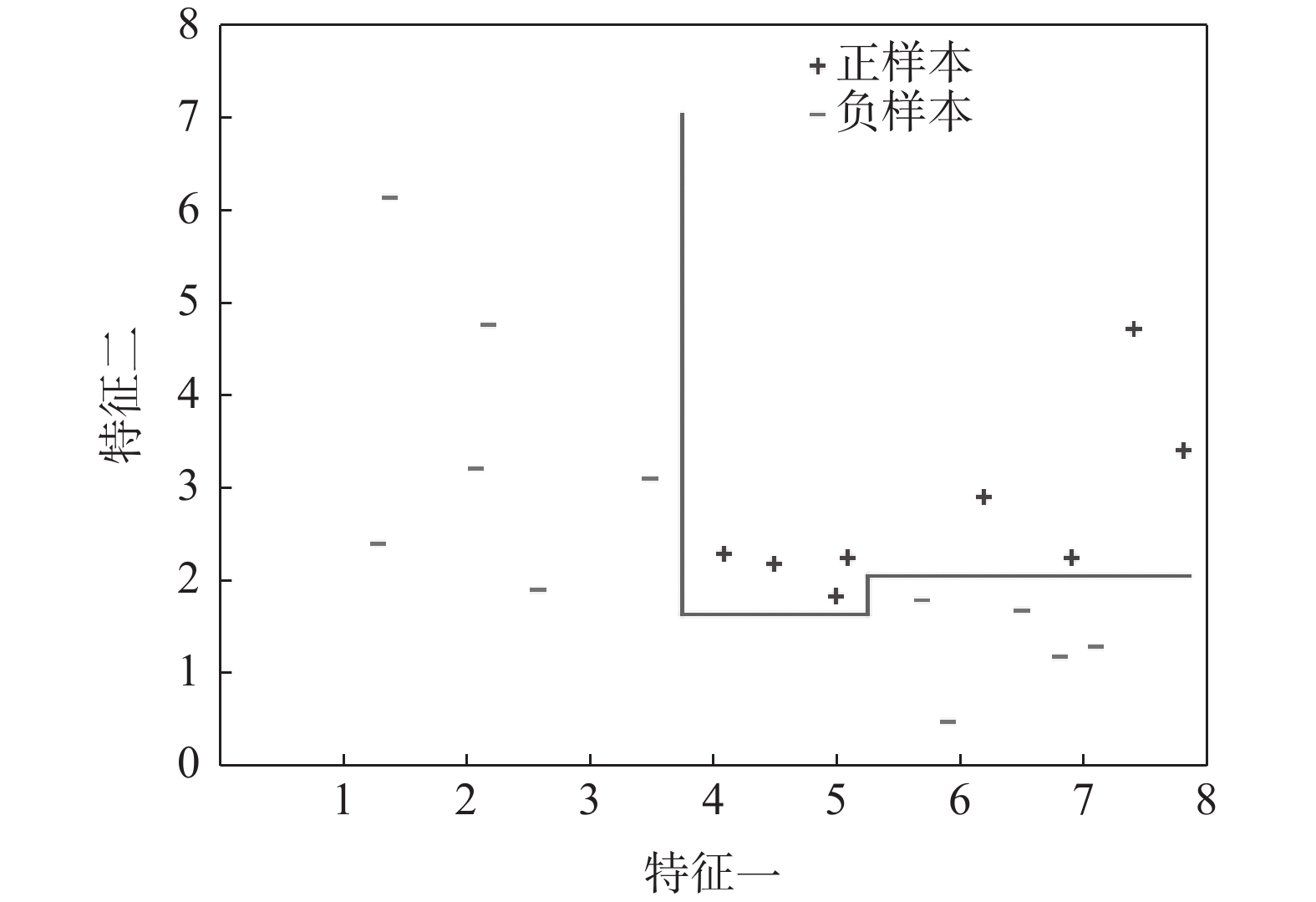
摘要: 运用决策树算法和随机森林算法来构建针对固态超离子导体的筛选模型。基于从文献收集的数据集和20个基于材料晶格常数的参数,建立了两种决策树模型、一种随机森林模型和一种作为对比的逻辑回归模型。通过对比,随机森林模型展示出较低的算法复杂度和较好的泛化能力。这些训练好的模型随后被用于筛选Material Project数据库中的含锂的化合物。随机森林模型的筛选结果将候选材料总数降低了87.76%,其中包含有数种已知的超离子导体材料,因而展现出了该模型的可靠性和高效性。所使用的模型建立方法可以显著减少搜寻理想物理属性的材料所需要的时间,从而加速了新材料的研发过程。Abstract: We here present a new approach of model construction to search for solid superionic materials in database by using decision tree and random forest algorithms. Based on a data set collected from literature and 20 features computed from lattice parameters, we constructed two decision tree models and a random forest model, as well as a logistic regression model for contrast. In comparison, the random forest model shows low algorithm complexity and better generalization ability. The well-trained models are then used to screen lithium-containing compounds in the material project database. Screening results of the random forest model reduce the candidate materials by 87.76% and consist of several known superionic materials, which exhibits efficiency and effectiveness of the model. The methodology of model building introduced here can remarkably reduce the searching range of materials with desired properties and thus accelerates the development of new materials.
-
表 1 随机森林模型与逻辑回归模筛选结果对比
Table 1. Screening results comparison of the random forest model and logistic regression model
Random forest, 200 trees Logistic regression Li3As1H36Se4N12 Li3As1H36Se4N12 Cs3Li3H12N6 Cs3Li3H12N6 Rb12Li2Nd22Se24Cl32O72 Rb12Li2Nd22Se24Cl32O72 Li1Ca4B3N6 Li1Ca4B3N6 Cs4Li2In2Cl12 Cs4Li2In2Cl12 Cs2Li1Al3F12 Cs2Li1Al3F12 Ba4Li1Sb3O12 Ba4Li1Sb3O12 K4Li2Al2F12 K4Li2Al2F12 Rb4Li2Ga2F12 Rb4Li2Ga2F12 Sr4Li1B3N6 Sr4Li1B3N6 Li9Er3Cl18 Li9Er3Cl18 Cs4Li6Ga2O8 Cs4Li6Ga2O8 Rb12Li2Pr22Se24Cl32O72 Rb12Li2Pr22Se24Cl32O72 Li2H6O4 Na8Li12Ga4O16 Li12Gd4B8O24 K2Li2Si4O10 Li2H12Br2O14 Sr8Li4C4Br12N8 Li1Sb1F6 Li1Er1Se2 Li1As1F6 Li4In4I16 K8Li32Al8O32 Li1Dy1Se2 Cs16Li8Si24O60 Li1Ho1Se2 Li2Si1Sn1S4 Rb2Li2S2 Li6U1O6 K20Li4Ge8O28 Li1P1F6 Li40Al8O32 Li6Bi2O8 Li1Er1S2 Li4Er4O8 Li1Tb1Se2 Li2Tm2O4 Li40Ga8O32 K1Li6Bi1O6 Li18In6Cl36 Li2Mg2B6H36N4 K8Li4B4P8O32 Li4Tm4Si4O16 Sr4Li16Ca4Si8O32 Rb4Li4Si2O8 Li8Te4O12 Li2In2O4 Li4Ga4Br16 Li4Ca12Si8N20 Li4Ga4I16 Sr2Li2Pr2Te2O12 K4Li2B2O6 Li4Ho4O8 Li2Sn4P10O30 K2Li6Pb2O8 Li4Ca36Mg4P28O112 Li1H1F2 Na12Li12In8F48 Li2Ge1Pb1S4 Li6Er2Br12 Li4Ca2Mg1Si2N6 Li2La4Sb2O12 Li2Sm2S4 Li1Ho1S2 Li2Ca1Si1O4 Na12Li12Al8F48 Li2Sm2Se4 Li1Dy1S2 Li2Ca1Ge1O4 Ba4Li4B4S12 -
[1] Holdren J P. Materials genome iniative for global competitiveness[M]. Washington, DC: NSTC, 2011. [2] Liu Q, Peng B, Shen M, et al. Polymer chain diffusion and Li+ hopping of poly(ethylene oxide)/LiAsF6 crystalline polymer electrolytes as studied by solid state NMR and ac impedance[J]. Solid State Ionics, 2014,255:74−79. doi: 10.1016/j.ssi.2013.11.053 [3] Tomita Y, Matsushita H, Kobayashi K, et al. Substitution effect of ionic conductivity in lithium ion conductor, Li3InBr6-xClx[J]. Solid State Ionics, 2008,179:867−870. doi: 10.1016/j.ssi.2008.02.012 [4] Wang J, Cheng C, Altukhov O, et al. Supramolecular functionalities influence the thermal properties: Interactions and conductivity behavior of poly(ethylene glycol)/LiAsF6 blends[J]. Polymers, 2013,5(3):937−953. doi: 10.3390/polym5030937 [5] WangY, Richards W D, Ong S P, et al. Design principles for solid-state lithium superionic conductors[J]. Nat. Mater., 2015,14(10):1026-1031. doi: 10.1038/NMAT4369 [6] Aravindan V, Gnanaraj J, Madhavi S, et al. Lithium-ion conducting electrolyte salts for lithium batteries[J]. Chem. -Eur. J., 2011,17(15):14326−14346. doi: 10.1002/chem.201101486 [7] Kamaya N, Homma K, Yamakawa Y, et al. A lithium superionic conductor[J]. Nat. Mater., 2011,10(9):682−686. doi: 10.1038/NMAT3066 [8] Hayashi A , Minami K , Mizuno F , et al. Formation of Li+ superionic crystals from the Li2S-P2S5 melt-quenched glasses[J]. J. Mater. Sci. , 2008 , 43: 1885-1889. [9] Xiang X D, Sun X, Briceno G, et al. A combinatorial approach to materials discovery[J]. Science, 1995,268(5218):1738−1740. doi: 10.1126/science.268.5218.1738 [10] Fujimura K, Seko A, Koyama Y, et al. Accelerated materials design of lithium superionic conductors based on first-principles calculations and machine learning algorithms[J]. Adv. Energy. Mater., 2013,3(8):980−985. doi: 10.1002/aenm.201300060 [11] Gao J, Chu G, He M, et al. Screening possible solid electrolytes by calculating the conduction pathways using bond valence method[J]. Sci. China Phys. Mech., 2014,57(8):1526−1535. doi: 10.1007/s11433-014-5511-4 [12] Sendek A D, Yang Q, Cubuk E D, et al. Holistic computational structure screening of more than 12 000 candidates for solid lithium-ion conductor materials[J]. Energ. Environ. Sci., 2017,10(1):306−320. doi: 10.1039/c6ee02697d [13] Zhai X, Chen M, Lu W. Accelerated search for perovskite materials with higher curie temperature based on the machine learning methods[J]. Comput. Mater. Sci., 2018,151:41−48. [14] Brodley C E, Utgoff P E. Multivariate decision trees[J]. Mach. Learn., 1995,19(1):45−77. doi: 10.1023/A:1022607123649 [15] Quinlan J R. Induction of decision trees[J]. Mach. Learn., 1986,1(1):81−106. [16] Breiman L. Random forests[J]. Mach. Learn., 2001,45(1):5−32. doi: 10.1023/A:1010933404324 [17] Breiman L. Using iterated bagging to debias regressions[J]. Mach. Learn., 2001,45(3):261−277. doi: 10.1023/A:1017934522171 [18] Yamada K, Kumano K, Okuda T. Lithium superionic conductors Li3InBr6 and LiInBr4 studied by 7Li, 115In NMR[J]. Solid State Ionics, 2006,177:1691−1695. doi: 10.1016/j.ssi.2006.06.026 [19] Stoeva Z, Martin-Litas I, Staunton E, et al. Ionic conductivity in the crystalline polymer electrolytes PEO6: LiXF6, X = P, As, Sb[J]. J. Am. Chem. Soc., 2003,125(15):4619−4626. doi: 10.1021/ja029326t [20] Yang H, Zhuang G V, Ross Jr. P N. Thermal stability of LiPF6 salt and Li-ion battery electrolytes containing LiPF6[J]. J. Power Sources, 2006,161(1):573−579. doi: 10.1016/j.jpowsour.2006.03.058 [21] York S S , Buckner M , Frech R. Ion-polymer and ion-ion interactions in linear poly(ethylenimine) complexed with LiCF3SO3 and LiSbF6[J]. Macromolecules, 2004, 37 (3): 994-999. doi: 10.1021/ma030478 y. [22] Yaroslavtseva T V , Bushkova O V. Glass transitions and ionic conductivity in a poly(butadiene-acrylonitrile)–LiAsF6 system[J]. Electrochim. Acta, 2011, 57: 212-219. -




 下载:
下载: