Electrocatalytic hydrogen production performance of MXene/cobalt phosphide composites
-
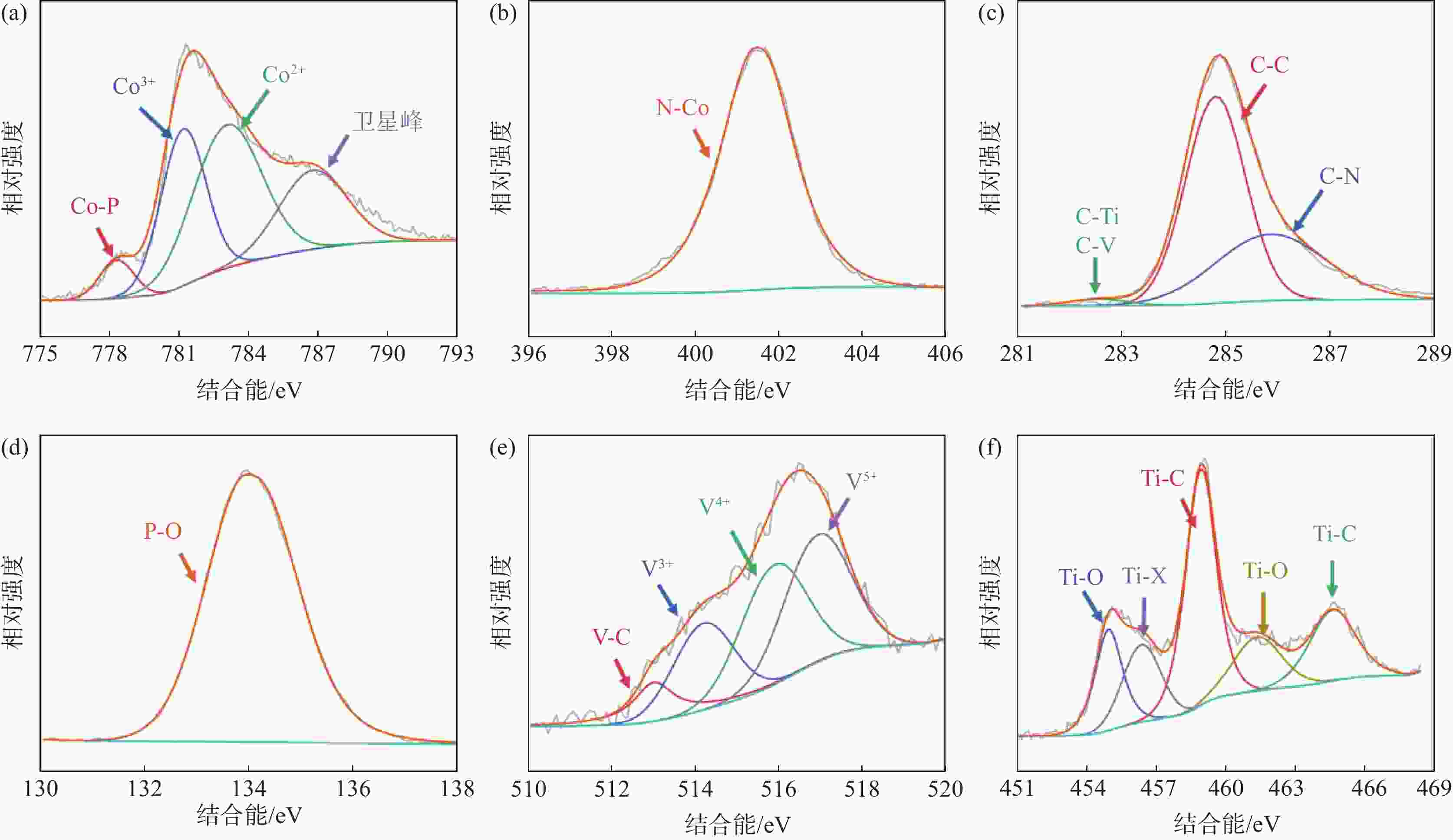
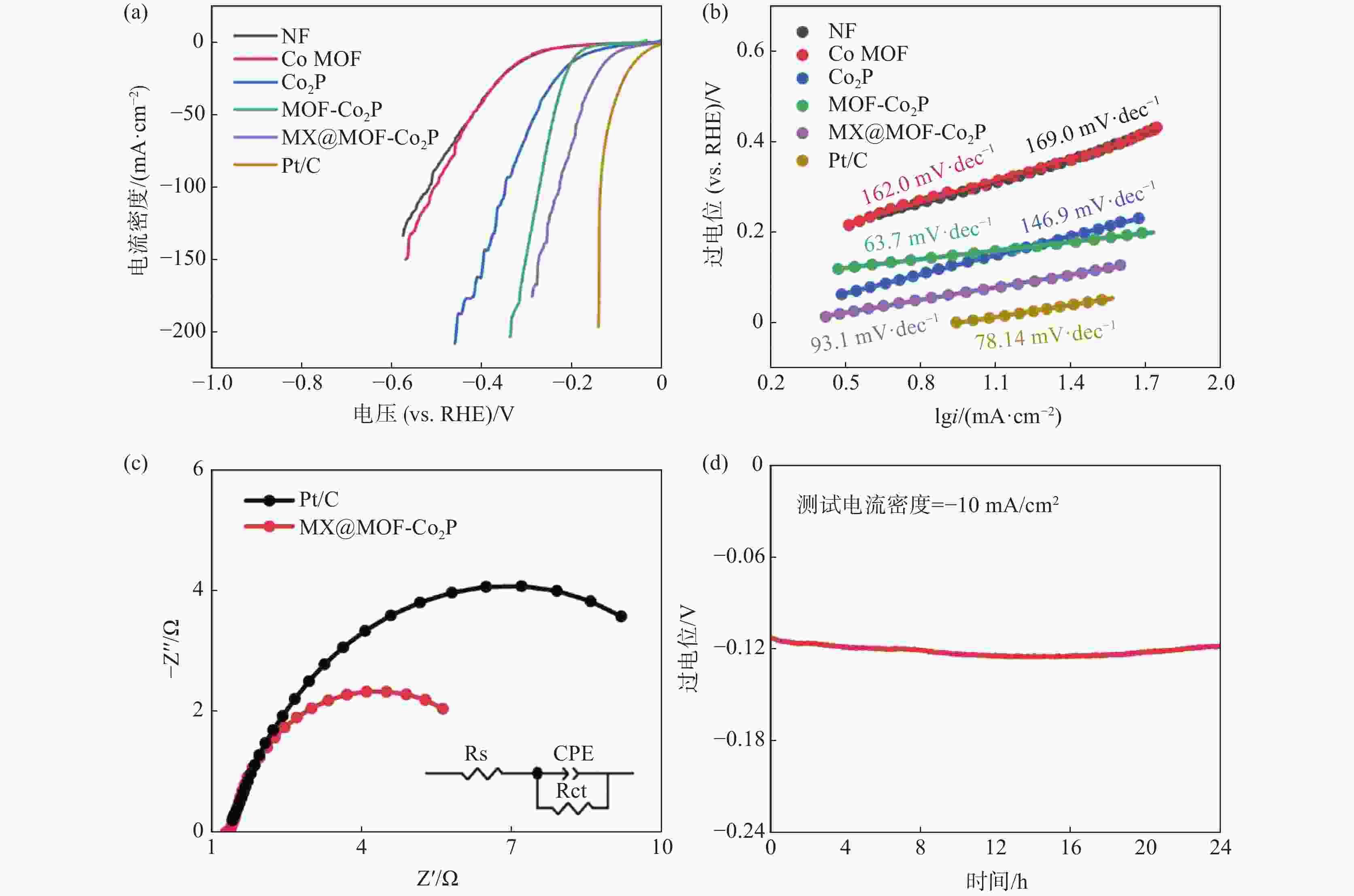
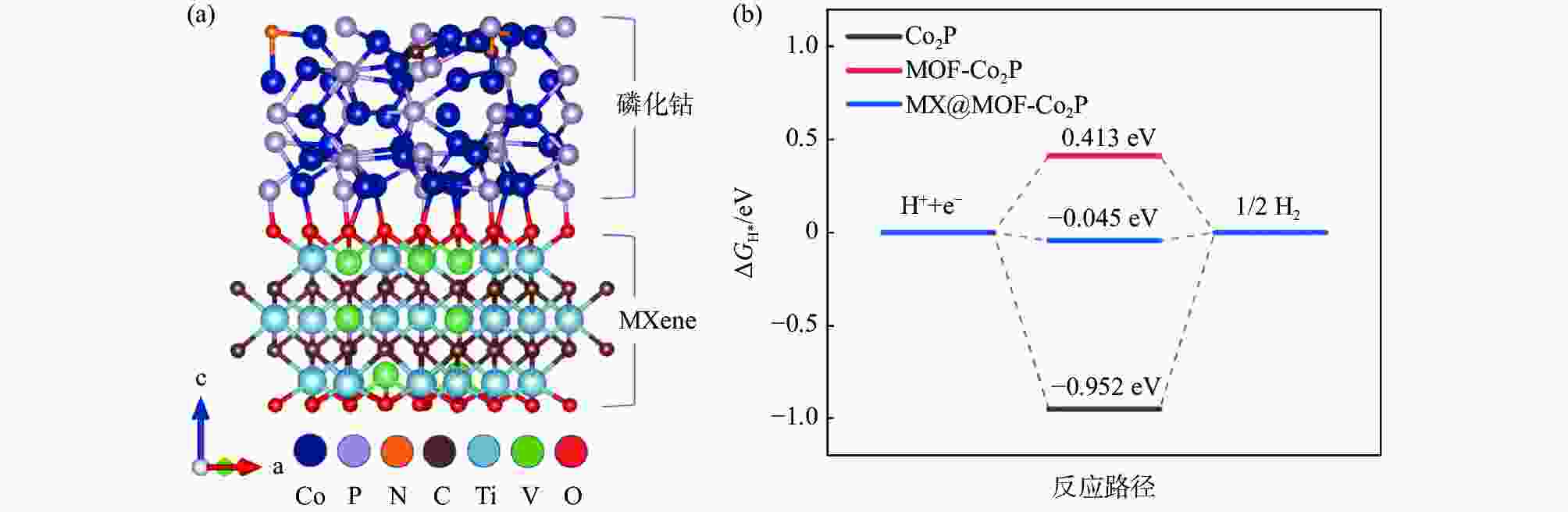
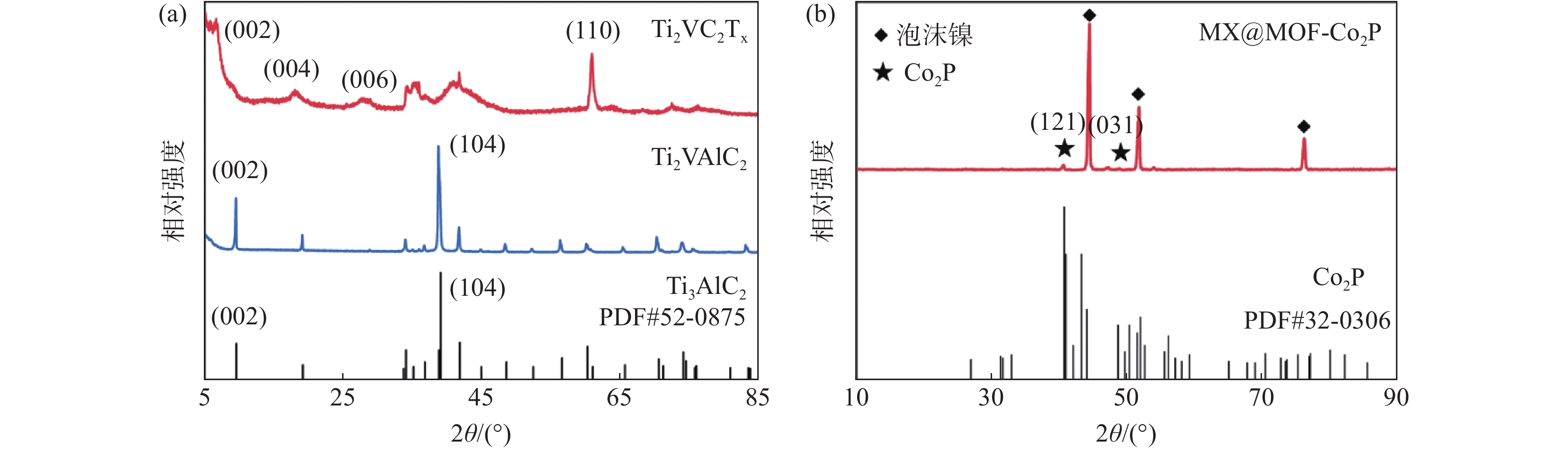
摘要: 针对传统电催化剂材料导电性差、活性位点易堵塞等问题,采用高导电性和优异亲水性的二维碳化钛钒(MXene)作为催化剂负载基底,在此基础上与具有高比表面积的金属有机框架衍生的磷化钴结合制备电解水制氢阴极材料。利用X射线衍射仪(XRD)、X射线光电子能谱(XPS)、扫描电子显微镜(SEM)、透射电子显微镜(TEM)、密度泛函理论计算(DFT)和电化学测试等手段,研究了复合电极材料的晶体结构、表面形貌特征、活性位点电子结构和电化学特性。结果表明,有机配体中的碳氮元素能有效调节电极材料中活性位点的电子结构,从而改善吸脱附动力学条件;二维MXene进一步降低了电极材料电荷转移电阻,所制备的催化剂在10 mA/cm2电流密度时仅需要114 mV的过电位,且长时间工作后依然保持稳定。研究结果为制备高性能非贵金属析氢催化剂提供了参考和思路。Abstract: To address the issues of low electrical conductivity and easy clogging of active sites in conventional electrocatalyst materials, a highly conductive and hydrophilic two-dimensional (2D) titanium vanadium carbide (MXene) is used as the catalyst loading substrate via combining metal organic framework derived cobalt phosphides with high specific surface areas to prepare cathode materials for electrolytic water splitting. The crystal structure, surface morphological characteristics, electronic structure of active sites, and electrochemical properties of the electrode materials are investigated by utilizing the X-ray diffractometer (XRD), X-ray photoelectron spectroscopy (XPS), scanning electron microscopy (SEM), transmission electron microscopy (TEM), density functional theory calculations (DFT), and electrochemical measurements. The results demonstrate that the carbon and nitrogen components of the organic ligands can effectively tune the electronic structure of the active sites and enhance the adsorption-desorption kinetics; the 2D MXene can further reduce the charge transfer resistance of the electrode materials, the as-obtained catalyst only requires 114 mV overpotential to achieve 10 mA/cm2 current density and exhibits outstanding stability. The research provides a reference for the preparation of high performance non-precious metal catalysts for hydrogen evolution reaction.
-
表 1 试验原料规格与厂家
Table 1. Specifications and manufacturers of raw materials
试剂名称 纯度/% 生产厂家 试剂名称 纯度/% 生产厂家 钛粉 99 阿拉丁 N,N-二甲基甲酰胺 99.50 阿拉丁 钒粉 99 阿拉丁 氢氧化钾 85 阿拉丁 铝粉 99 阿拉丁 次亚磷酸钠 98 麦克林 石墨粉 99 阿拉丁 二甲基咪唑 99 麦克林 氢氟酸 40~49 麦克林 硝酸钴六水合物 99 麦克林 盐酸 37 麦克林 全氟化树脂 5 阿达玛斯 四甲基氢氧化铵 99.70 麦克林 20% Pt/C粉末 20 麦克林 无水乙醇 95 川东化工 去离子水 分析纯 自制 -
[1] Benck J, Hellstern J, Kibsgaard P, et al. Catalyzing the hydrogen evolution reaction (HER) with molybdenum sulfide nanomaterials[J]. ACS Catal, 2014,4:3957−3971. doi: 10.1021/cs500923c [2] Cao L M, Zhang J, Ding L W, et al. Metal-organic frameworks derived transition metal phosphides for electrocatalytic water splitting[J]. J Energy Chem, 2022,68:494−520. doi: 10.1016/j.jechem.2021.12.006 [3] Bavykina A, Kolobov N, Khan I S, et al. Metal-organic frameworks in heterogeneous catalysis: recent progress, new trends, and future perspectives[J]. Chem Rev, 2020,120:8468−8535. [4] Liu T, Li P, Yao N, et al. CoP-doped mof-based electrocatalyst for ph-universal hydrogen evolution reaction[J]. Angew Chem Int Ed, 2019,58:4679−4684. [5] Wang P C, Xu Z A, Lin Y Q, et al. MOF-derived N-doped CoP and Fe-doped CoOOH ultrathin nanosheets electrocatalysts for overall water splitting[J]. ACS Sustain Chem, 2020,8:8949−8957. [6] Naguib M, Mochalin V N, Barsoum M W, et al. 25th anniversary article: mxenes: a new family of two-dimensional materials[J]. Adv Mater, 2014,26:992−1005. doi: 10.1002/adma.201304138 [7] Li J Z, Chen C, Lv Z P, et al. Constructing heterostructures of ZIF-67 derived C, N doped Co2P and Ti2VC2Tx mxene for enhanced OER[J]. J Mater Sci Technol, 2023,145:74−82. doi: 10.1016/j.jmst.2022.10.048 [8] Cai G R, Yan P, Zhang L L, et al. Metal–organic framework-based hierarchically porous materials: synthesis and applications[J]. Chem Rev, 2021,121:12278−12326. doi: 10.1021/acs.chemrev.1c00243 [9] Jian K L, Ma W S, Lv Z P, et al. Tuning the electronic structure of the CoP/Ni2P nanostructure by nitrogen doping for an efficient hydrogen evolution reaction in alkaline media[J]. Inorg Chem, 2021,60:18544−18552. doi: 10.1021/acs.inorgchem.1c03145 [10] Wang M, Ma W S, Lv Z P, et al. Co-doped Ni3N nanosheets with electron redistribution as bifunctional electrocatalysts for efficient water splitting[J]. J Phys Chem Lett, 2021,12:1581−1587. doi: 10.1021/acs.jpclett.0c03804 [11] Lv Z P, Ma W S, Dang J, et al. Induction of Co2P growth on a MXene (Ti3C2Tx)-Modified self-supporting electrode for efficient overall water splitting[J]. J Phys Chem Lett, 2021,12:4841−4848. doi: 10.1021/acs.jpclett.1c01345 [12] Zou Z H, Wang J L, Pan H R, et al. Enhanced oxygen evolution reaction of defective CoP/MOF-integrated electrocatalyst by partial phosphating[J]. J Mater Chem A, 2020,8:14099−14105. doi: 10.1039/D0TA04025H [13] He Z Q, Rong T D, Li Y, et al. Two-dimensional TiVC solid-solution MXene as surface-enhanced raman scattering substrate[J]. ACS Nano, 2022,16:4072−4083. doi: 10.1021/acsnano.1c09736 [14] Lv Z P, Ma W S, Wang M, et al. Co-constructing interfaces of multiheterostructure on MXene (Ti3C2Tx)-modified 3D self-supporting electrode for ultraefficient electrocatalytic HER in alkaline media[J]. Adv Funct Mater, 2021,31:2102576. doi: 10.1002/adfm.202102576 [15] Zhang C L, Xie Y, Liu J T, et al. 1D core−shell MOFs derived CoP nanoparticles-embedded N-doped porous carbon nanotubes anchored with MoS2 nanosheets as efficient bifunctional electrocatalysts[J]. Chem Eng J, 2021,419:129977. doi: 10.1016/j.cej.2021.129977 [16] Nørskov J K, Bligaard T, Logadottir A, et al. Trends in the exchange current for hydrogen evolution[J]. J Electrochem Soc, 2005,152:J23−J26. doi: 10.1149/1.1856988 -





 下载:
下载: