Phase transformation and electrical resistivity of reduced ilmenite concentrate carbon-containing pellets
-
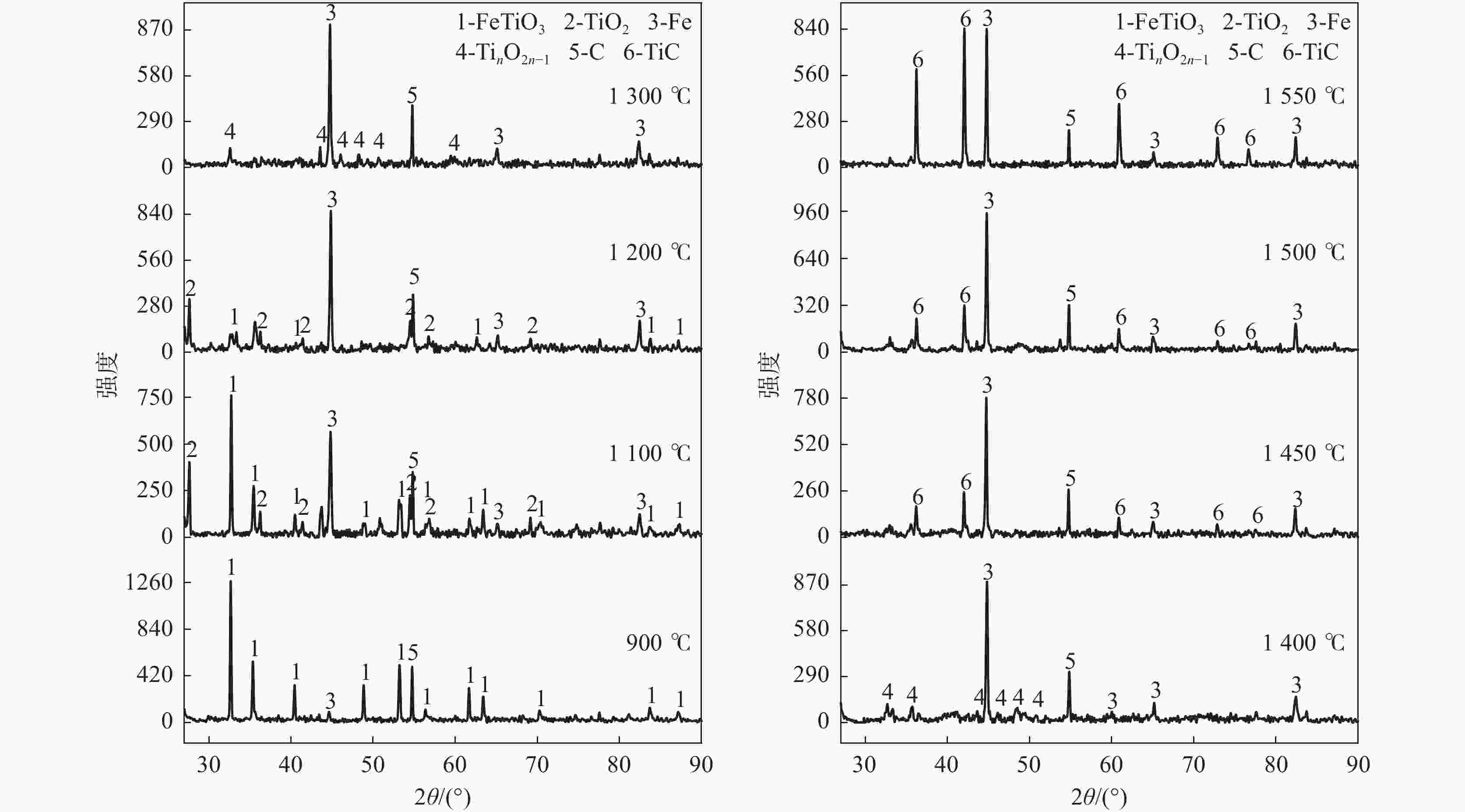
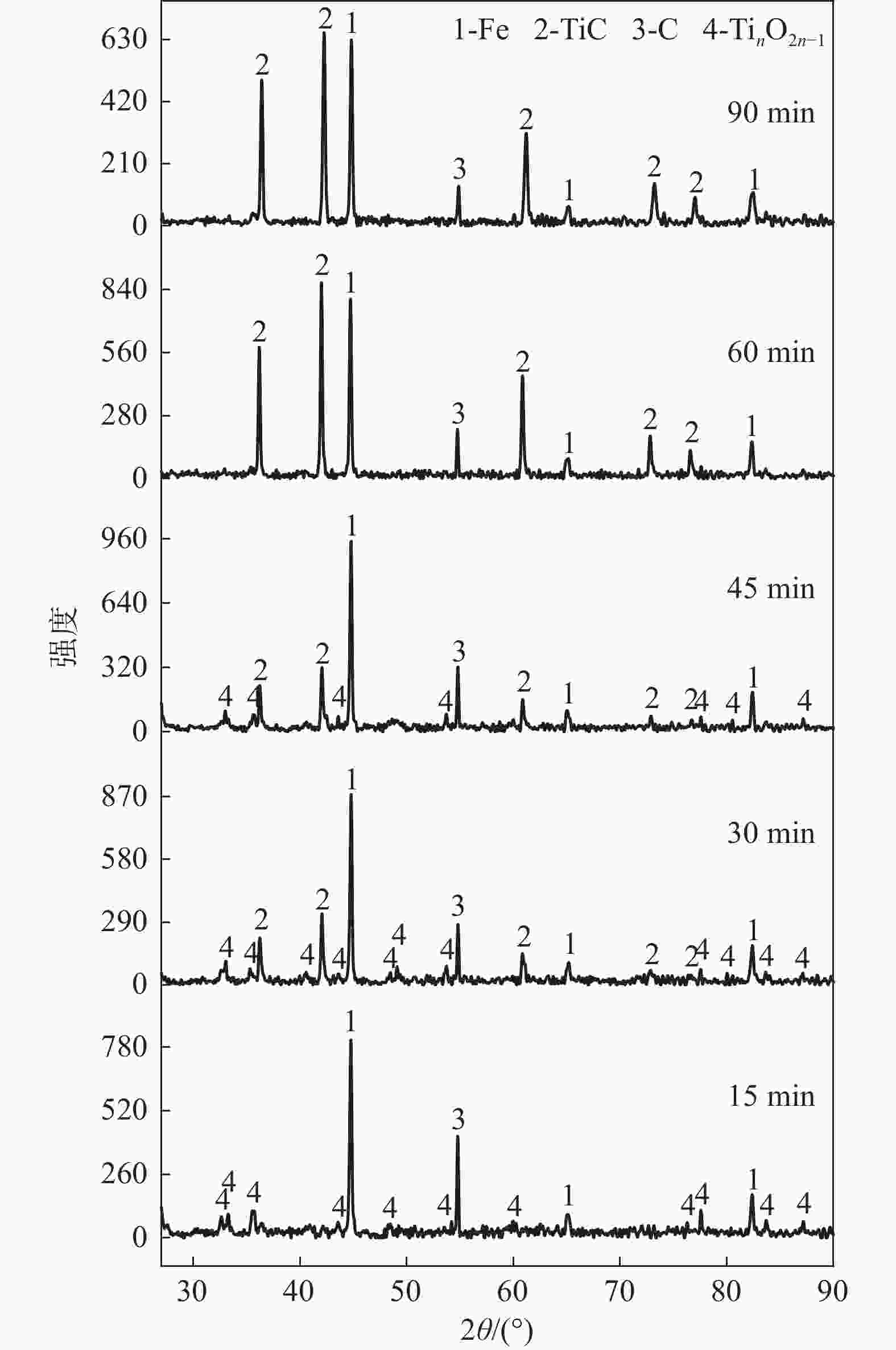
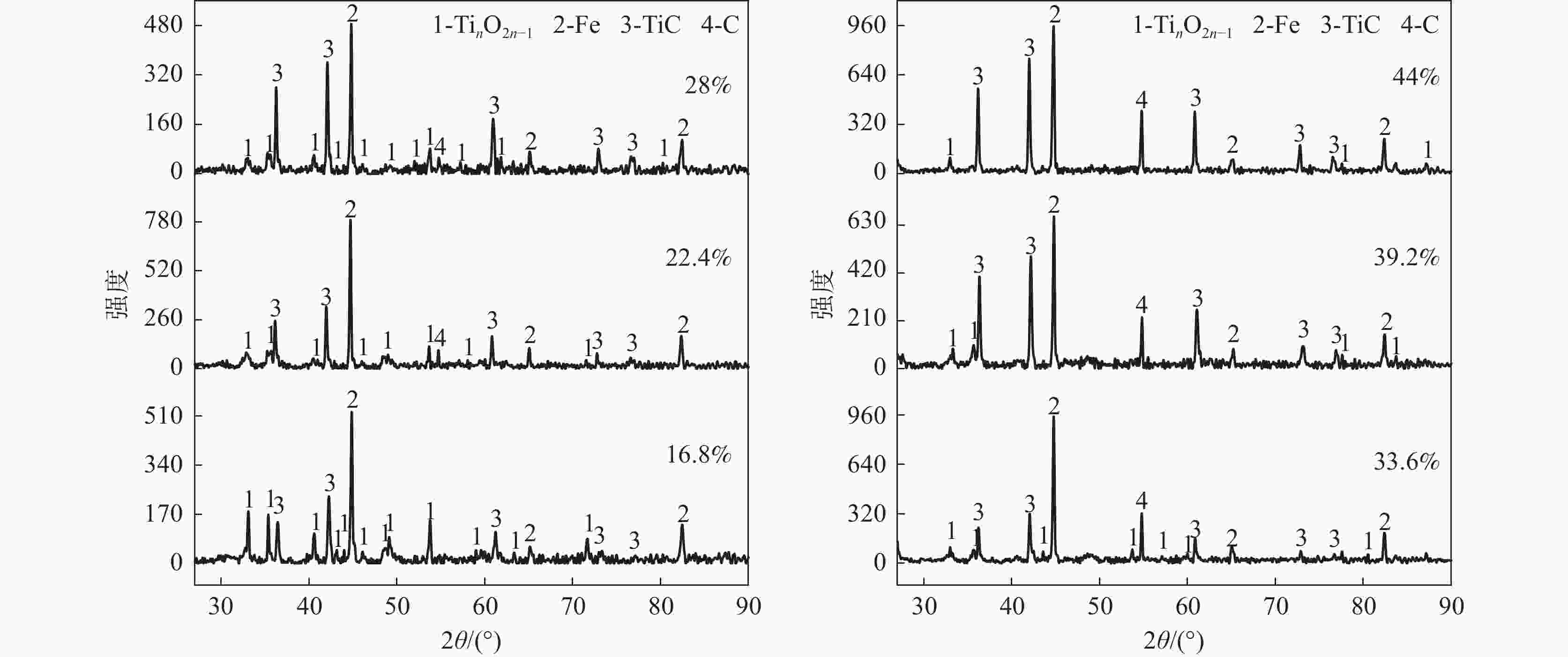
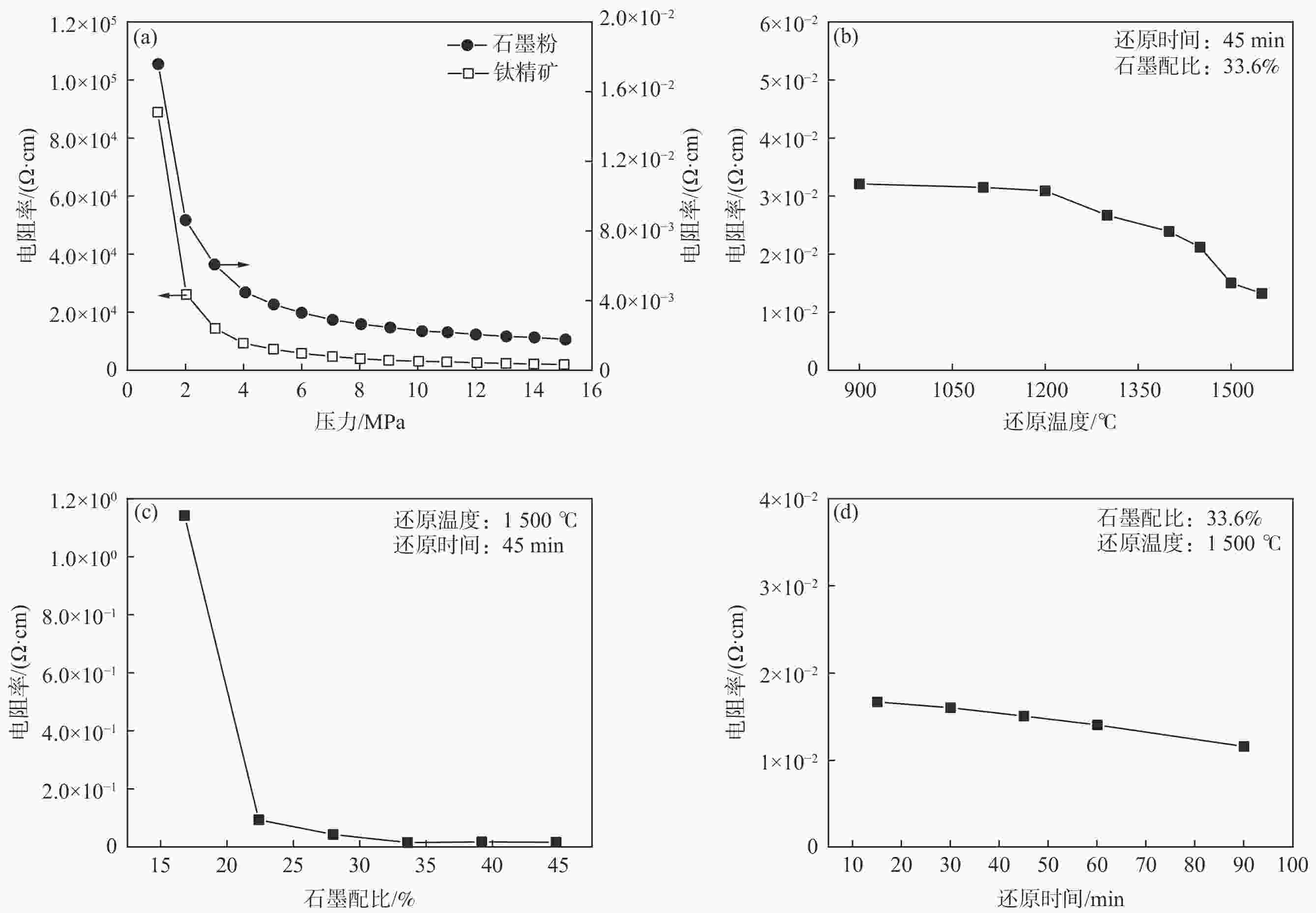
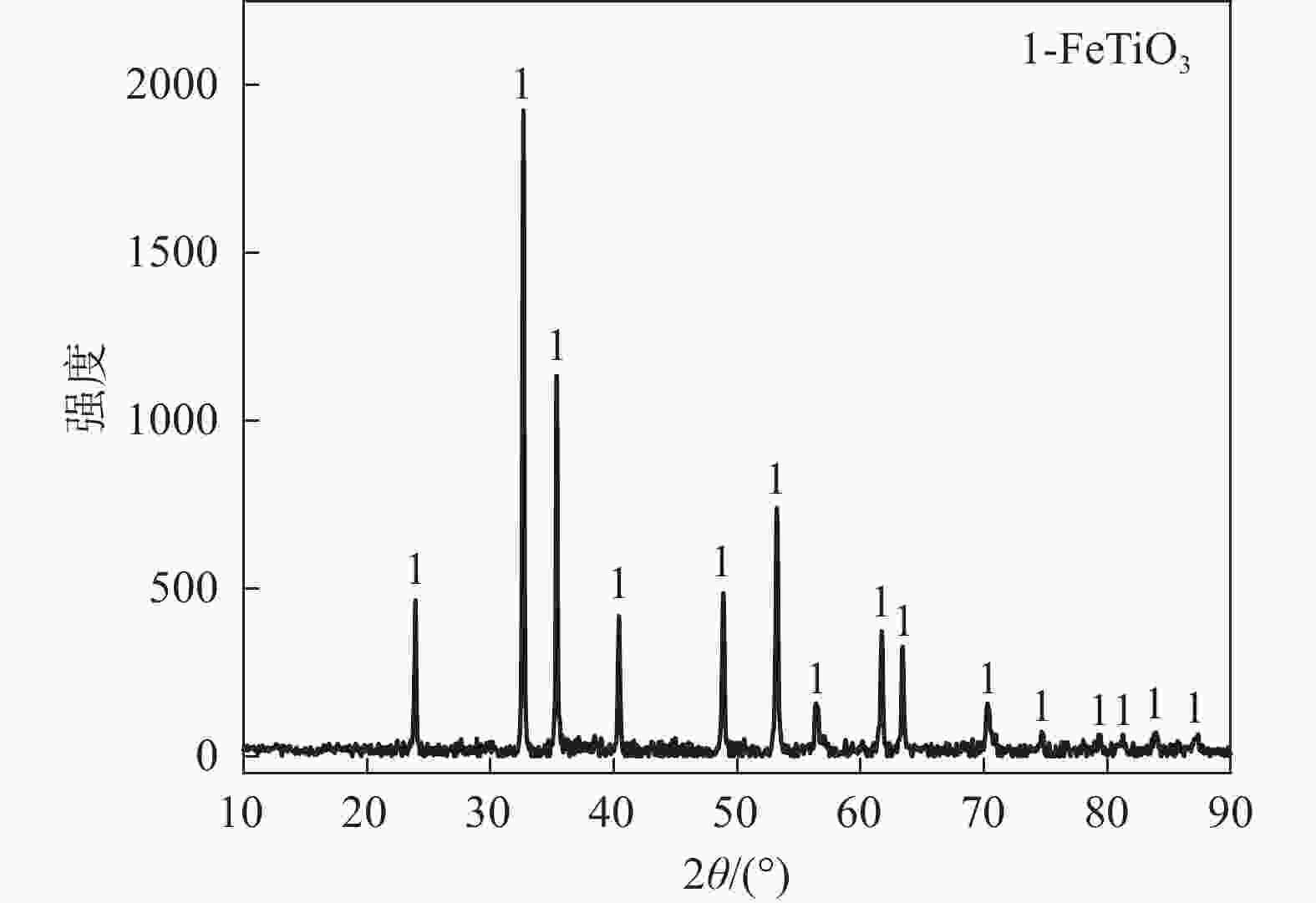
摘要: 以石墨粉为还原剂,研究钛精矿内配碳球团还原过程中的物相转变规律和还原产物的电阻率。结果表明,钛精矿内配碳球团的还原过程先后主要经历铁氧化物的还原和钛氧化物的还原两个过程,且提高还原温度、延长还原时间和增加石墨配比均有利于钛氧化物还原程度的加深;在还原时间为60 min,石墨配比为33.6%的条件下,还原温度由900 ℃提高到1550 ℃时,还原产物物相随温度升高的转变过程为:FeTiO3→Fe+TiO2→Fe+TinO2n−1(n≈1,2,3,4)→Fe+TiC。还原产物电阻率测试结果表明,还原温度和石墨配比对还原产物电阻率影响较大,还原时间影响较小;在石墨配比为33.6%,还原温度1300 ℃,还原时间为45 min的条件下,还原产物的电阻率的值为2.67×10−2 Ω·cm。Abstract: Using the graphite powder as a reducing agent, the phase transformation and electrical resistivity of the reduced products during the reduction process of carbon-containing ilmenite concentrate pellets were studied. The XRD analysis results indicate that the reduction process of carbon-containing ilmenite concentrate pellets mainly undergoes two processes: the reduction of iron oxide and the reduction of titanium oxide. Increasing the reduction temperature, prolonging the reduction time, and increasing the graphite ratio are all beneficial in deepening the reduction degree of titanium oxide. With a reduction time of 60 min and a graphite ratio of 33.6%, the phase transformation process of the reduction products with increased reduction temperature from 900 ℃ to 1550 ℃ was as follows: FeTiO3→Fe+TiO2→Fe+TinO2n−1(n≈1,2,3,4)→Fe+TiC. The electrical resistivity test results of the reduction products show that the reduction temperature and graphite ratio have a significant impact on the electrical resistivity of the reduced products, while the reduction time has a small impact. Under the conditions of graphite ratio of 33.6%, reduction temperature of 1300 ℃, and reduction time of 45 min, the electrical resistivity of the reduced product was 2.67 × 10−2 Ω·cm.
-
表 1 钛精矿的主要化学成分
Table 1. Main chemical composition of ilmenite concentrate
% TiO2 FeO CaO MgO SiO2 Al2O3 P2O5 SO3 K2O Cr2O3 MnO 46.70 42.03 0.59 5.13 2.26 1.00 0.10 0.65 0.03 0.15 0.77 -
[1] 杨绍利. 钒钛材料[M]. 北京: 冶金工业出版社, 2007.Yang Shaoli. Vanadium titanium material[M]. Beijing: Metallurgical Industry Press, 2007. [2] Jia Hong, Lu Fusheng, Hao Bin. China titanium industry development report 2018[J]. Iron Steel Vanadium Titanium, 2019,40(3):158−163. (贾翃, 逯福生, 郝斌. 2018年中国钛工业发展报告[J]. 钢铁钒钛, 2019,40(3):158−163. doi: 10.7513/j.issn.1004-7638.2019.03.028Jia Hong, Lu Fusheng, Hao Bin. China titanium industry development report 2018[J]. Iron Steel Vanadium Titanium, 2019, 40(3): 158-163. doi: 10.7513/j.issn.1004-7638.2019.03.028 [3] Zhang Li, Hu Huiping, Wei Liangping, et al. Hydrochloric acid leaching behaviour of mechanically activated Panxi ilmenite (FeTiO3)[J]. Separation & Purification Technology, 2010,73(2):173−178. [4] Zhang Li, Hu Huiping, Liao Zhi, et al. Hydrochloric acid leaching behavior of different treated Panxi ilmenite concentrations[J]. Hydrometallurgy, 2011,107(1-2):40−47. doi: 10.1016/j.hydromet.2011.01.006 [5] Song Bing, Lü Xuewei, Xu Jian, et al. Effect of wet grinding on carbothermic reduction of ilmenite concentrate[J]. International Journal of Mineral Processing, 2015,142:101−106. doi: 10.1016/j.minpro.2015.02.014 [6] Lü Xiangdong, Huang Run, Wu Qinzhi, et al. Non-isothermal reduction kinetics during vacuum carbothermal reduction of ilmenite concentrate[J]. Vacuum, 2019,160:139−145. doi: 10.1016/j.vacuum.2018.11.009 [7] Lü Wei, Bai Chenguang, Lü Xuewei, et al. Carbothermic reduction of ilmenite concentrate in semi-molten state by adding sodium sulfate[J]. Powder Technology, 2018,340:354−361. doi: 10.1016/j.powtec.2018.09.043 [8] Liao Xuefeng, Peng Jinhui, Zhang Libo, et al. Enhanced carbothermic reduction of ilmenite placer by additional ferrosilicon[J]. Journal of Alloys and Compounds, 2017,708:1110−1116. doi: 10.1016/j.jallcom.2017.03.113 [9] Zhang Guohua, Gou Haipeng, Wu Kehan, et al. Carbothermic reduction of Panzhihua ilmenite in vacuum[J]. Vacuum, 2017,143:199−208. doi: 10.1016/j.vacuum.2017.06.016 [10] Han Kexi. Analysis on electricity consumption for titanium slag smelting with prereduced concentrate pellets[J]. Iron Steel Vanadium Titanium, 2014,35(2):51−55. (韩可喜. 钛精矿预还原球团冶炼钛渣的电耗水平分析[J]. 钢铁钒钛, 2014,35(2):51−55. doi: 10.7513/j.issn.1004-7638.2014.02.010Han Kexi. Analysis on electricity consumption for titanium slag smelting with prereduced concentrate pellets[J]. Iron Steel Vanadium Titanium, 2014, 35(2): 51-55. doi: 10.7513/j.issn.1004-7638.2014.02.010 [11] Chuan Xiuyun, Chen Daizhang, Zhou Xunruo, et al. The electrical property and its mechanism of intercalation compounds of CuCl2[J]. Acta Physica Sinica, 1999,48(6):159−164. (传秀云, 陈代璋, 周旬若, 等. CuCl2-石墨层间化合物导电性能及其机理研究[J]. 物理学报, 1999,48(6):159−164.Chuan Xiuyun, Chen Daizhang, Zhou Xunruo, et al. The electrical property and its mechanism of intercalation compounds of CuCl2[J]. Acta Physica Sinica, 1999, 48(6): 159-164. [12] Wu Enhui, Li Jun, Xu Zhong, et al. Physicochemical properties of metallized pellets of high-chromium vanadium-bearing titanomagnetite[J]. Iron & Steel, 2023,58(2):30−38. (吴恩辉, 李军, 徐众, 等. 高铬型钒钛铁精矿金属化球团的物化性能[J]. 钢铁, 2023,58(2):30−38. doi: 10.13228/j.boyuan.issn0449-749x.20220496Wu Enhui, Li Jun, Xu Zhong, et al. Physicochemical properties of metallized pellets of high-chromium vanadium-bearing titanomagnetite[J]. Iron & Steel, 2023, 58(2): 30-38. doi: 10.13228/j.boyuan.issn0449-749x.20220496 [13] Sen Wei, Xu Baoqiang, Yang Bin, et al. Research progress on preparation of TiC powders[J]. Light Metals, 2010,(12):44−48. (森维, 徐宝强, 杨斌, 等. 碳化钛粉末制备方法的研究进展[J]. 轻金属, 2010,(12):44−48. doi: 10.13662/j.cnki.qjs.2010.12.009Sen Wei, Xu Baoqiang, Yang Bin, et al. Research progress on preparation of TiC powders[J]. Light Metals, 2010 (12): 44-48. doi: 10.13662/j.cnki.qjs.2010.12.009 -




 下载:
下载: