Study on the transformation mechanism of chromium in chromium sludge under reducing atmosphere
-
摘要: 在还原气氛下对含铬污泥中铬形态转化机理进行了研究,并讨论了转底炉用于处理含铬污泥的可行性。结果表明,铬在污泥中主要以氢氧化铬、铬酸钙和铬酸钠的形式存在;在CO-CO2气氛下,还原后铬泥中的三价铬不会氧化成六价铬且铬泥原有的六价铬会被还原成三价铬,在相同CO浓度下,随着铬泥配比增大,六价铬转化率逐步降低;在CO-CO2-O2-N2气氛下,配加3%和6%的氧气后,六价铬转化率和六价铬变化率均为负值,三价铬仍不会氧化成六价铬。因此,将含铬污泥在转底炉工序进行无害化处置是可行的。Abstract: In this paper, the transformation mechanism of chromium in chromium sludge was studied under reducing atmosphere, and the feasibility of rotary hearth furnace (RHF) for treating chromium sludge was discussed. The results show that chromium mainly exists in the form of chromium hydroxide, calcium chromate and sodium chromate in the sludge. Under the CO-CO2 atmosphere, the Cr(III) in the chromium sludge will not be oxidized to Cr(VI) and the original hexavalent chromium in the sludge will be reduced to trivalent chromium. Under the same CO concentration, the conversion rate of hexavalent chromium gradually decreases with the proportion of chromium mud increase. Under the CO-CO2-O2-N2 atmosphere, after adding 3% and 6% oxygen, the conversion rate of hexavalent chromium and the change rate of hexavalent chromium are both negative, and Cr(III) will still not be oxidized to Cr(VI). Therefore, it is feasible to carry out harmless disposal of chromium sludge in the rotary hearth furnace process.
-
Key words:
- chromium sludge /
- transformation mechanism /
- rotary hearth furnace /
- reducing atmosphere
-
表 1 含铬污泥的主要化学成分
Table 1. Main chemical composition of chromium sludge
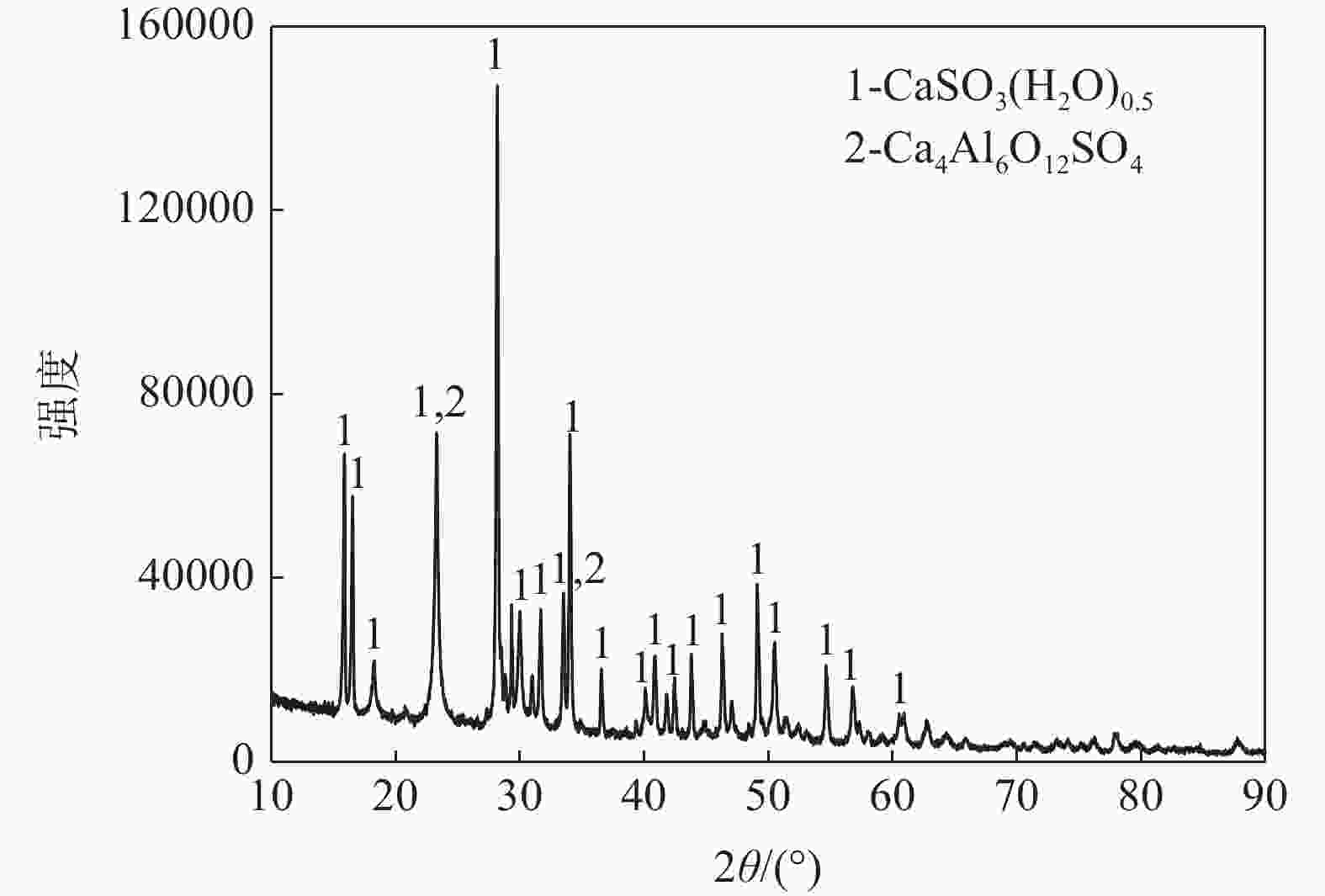
% Cr2O3 CaO Fe2O3 SiO2 Al2O3 MgO Cl Na2O K2O 5.937 32.50 1.781 0.759 9.86 0.165 0.178 0.219 0.012 CuO ZnO PbO TiO2 SrO Bi P2O5 SO3 其它 0.01 0.0447 0.009 0.0320 0.0123 0.0036 0.076 46.51 1.8914 表 2 含铁尘泥混合料的主要化学成分
Table 2. Main chemical composition of iron-bearing dust
% Cr2O3 CaO Fe2O3 SiO2 Al2O3 MgO Cl Na2O K2O 0.029 7.92 67.96 3.39 1.69 2.03 2.06 0.982 0.361 CuO ZnO PbO TiO2 SrO MnO P2O5 SO3 其它 0.022 2.319 0.258 0.0949 0.008 0.529 0.0202 1.28 9.04 表 3 试验焙烧制度
Table 3. Roasting parameters of the experiment
区域 温度/℃ 时间/min 排烟段 1050~1150 6.88 预热段 还原一区 1150~1230 5.00 还原二区 1230~1280 4.38 还原三区 1250 5.00 冷却段 1150 0.83 表 4 CO-CO2气氛下六价铬的变化情况
Table 4. Variation of hexavalent chromium content under CO-CO2 atmosphere
气氛 $ \phi $/% β/% 0.2% 1.0% 3.0% 5.0% 0.2% 1.0% 3.0% 5.0% O2=100% 0.56 0.56 0.24 0.26 11.75 29.12 24.57 50.60 CO=2% CO2=98% −2.18 −1.00 −0.49 −0.27 −46.06 −52.23 −49.08 −51.52 CO=5% CO2=95% −2.94 −0.94 −0.60 −0.21 −62.08 −49.01 −60.13 −41.68 CO=10% CO2=90% −1.15 −0.35 −0.35 −0.15 −44.24 −58.44 −45.73 −48.38 CO=20% CO2=80% −1.79 −0.94 −0.45 −0.17 −37.87 −48.96 −45.51 −33.33 表 5 混合气氛下六价铬的变化情况
Table 5. Variation of hexavalent chromium content under mixed atmosphere
气氛 $ \phi $/% β/% 0.2% 0.5% 1.0% 0.2% 0.5% 1.0% CO=10%,O2=3%,N2+CO2=87% −3.39 −7.47 −13.42 −38.65 −63.58 −77.37 CO=5%,O2=6%,N2+CO2=89% −3.84 −7.70 −13.20 −43.80 −65.59 −76.10 表 6 不同气氛下铬元素的分布情况
Table 6. Distribution of chromium element in different atmospheres
试验参数 ∆Cr(焙烧后-焙烧前) 气氛 铬泥配比/% ∆TCr/g ∆Cr6+/g ∆Cr3+/g CO=2%,
CO2=98%0.2 −0.0022 −0.0002 −0.0020 1.0 −0.0059 −0.0002 −0.0057 3.0 −0.0076 −0.0003 −0.0073 5.0 −0.0065 −0.0002 −0.0064 CO=5%,
CO2=95%0.2 −0.0016 −0.0004 −0.0013 1.0 −0.0066 −0.0002 −0.0063 3.0 −0.0066 −0.0003 −0.0063 5.0 −0.0078 −0.0002 −0.0076 CO=10%,
CO2=90%0.2 −0.0004 −0.0001 −0.0003 1.0 −0.0074 −0.0001 −0.0074 3.0 −0.0050 −0.0002 −0.0049 5.0 −0.0085 −0.0001 −0.0084 CO=20%,
CO2=80%0.2 −0.0007 −0.0002 −0.0005 1.0 −0.0073 −0.0002 −0.0070 3.0 −0.0008 −0.0002 −0.0006 5.0 −0.0083 −0.0001 −0.0082 CO=10%,O2=3%,
N2+CO2=87%0.2 −0.0017 −0.0005 −0.0012 0.5 −0.0039 −0.0012 −0.0027 1.0 −0.0038 −0.0022 0.0016 CO=5%,O2=6%,
N2+CO2=89%0.2 −0.0008 −0.0005 −0.0003 0.5 −0.0039 −0.0012 −0.0027 1.0 −0.0039 −0.0022 0.0017 -
[1] Na Xianzhao, Qi Yuanhong. Resourceful disposal of chromium-containing residues in ferrous metallurgical process[J]. Journal of Iron and Steel Research, 2009,21(4):1−4. (那贤昭, 齐渊洪. 铬渣在钢铁冶金过程中的资源化利用[J]. 钢铁研究学报, 2009,21(4):1−4. doi: 10.13228/j.boyuan.issn1001-0963.2009.04.010 [2] Shi Lei, Chen Ronghuan, Wang Ruyi. Application prospect of chromium sludge pellet in iron and steel industry[J]. Recyclable Resources and Circular Economy, 2007,(1):33−36. (石磊, 陈荣欢, 王如意. 含铬污泥球团在钢铁工业中的应用前景[J]. 再生资源研究, 2007,(1):33−36. [3] Zhang Lei, Liu Shangchao, Zhang Daoquan, et al. Application research on co-disposal of chromium sludge in the sintering and irongmaking[J]. Sintering and Pelletizing, 2018,43(5):61−64. (张垒, 刘尚超, 张道权, 等. 烧结炼铁协同处置含铬污泥的应用研究[J]. 烧结球团, 2018,43(5):61−64. doi: 10.13403/j.sjqt.2018.05.073 [4] Shi Lei, Chen Ronghuan, Wang Ruyi. Study on metallurgical properties and pellet proportioning technology of stainless steel cold-rolling sludge[J]. Baosteel Technology, 2013,(2):23−27. (石磊, 陈荣欢, 王如意. 不锈钢冷轧污泥配矿压球工艺及冶金性能研究[J]. 宝钢技术, 2013,(2):23−27. doi: 10.3969/j.issn.1008-0716.2013.02.005 [5] 范志功. 冷轧含铬废水的处理及含铬污泥回收利用的研究[D]. 武汉: 武汉科技大学, 2009.Fan Zhigong. Study on treatment of cold rolling wastewater containing chromium and recovery in sludge[D]. Wuhan: Wuhan University of Science and Technology, 2009. [6] Fang Duokui, Li Xianwei, Su Junjie. Analysis on research progress of cold-rolled chromium-containing sludge[J]. Resources Economization & Environmental Protection, 2013,(6):54. (房多奎, 李咸伟, 苏俊杰. 冷轧含铬污泥资源化途径研究进展分析[J]. 资源节约与环保, 2013,(6):54. doi: 10.3969/j.issn.1673-2251.2013.06.037 [7] Chen J, Jiao F, Zhang L, et al. Elucidating the mechanism of Cr (VI) formation upon the interaction with metal oxides during coal oxy-fuel combustion[J]. Journal of Hazardous Materials, 2013,261:260−268. doi: 10.1016/j.jhazmat.2013.07.023 [8] Chen J C, Wey M Y, Chiang B C, et al. The simulation of hexavalent chromium formation under various incineration conditions[J]. Chemosphere, 1998,36(7):1553−1564. doi: 10.1016/S0045-6535(97)10053-4 -




 下载:
下载:



