Extraction of vanadium from high-chromium vanadium-bearing titanomagnetite pellets by oxidation roasting-HCl leaching process
-
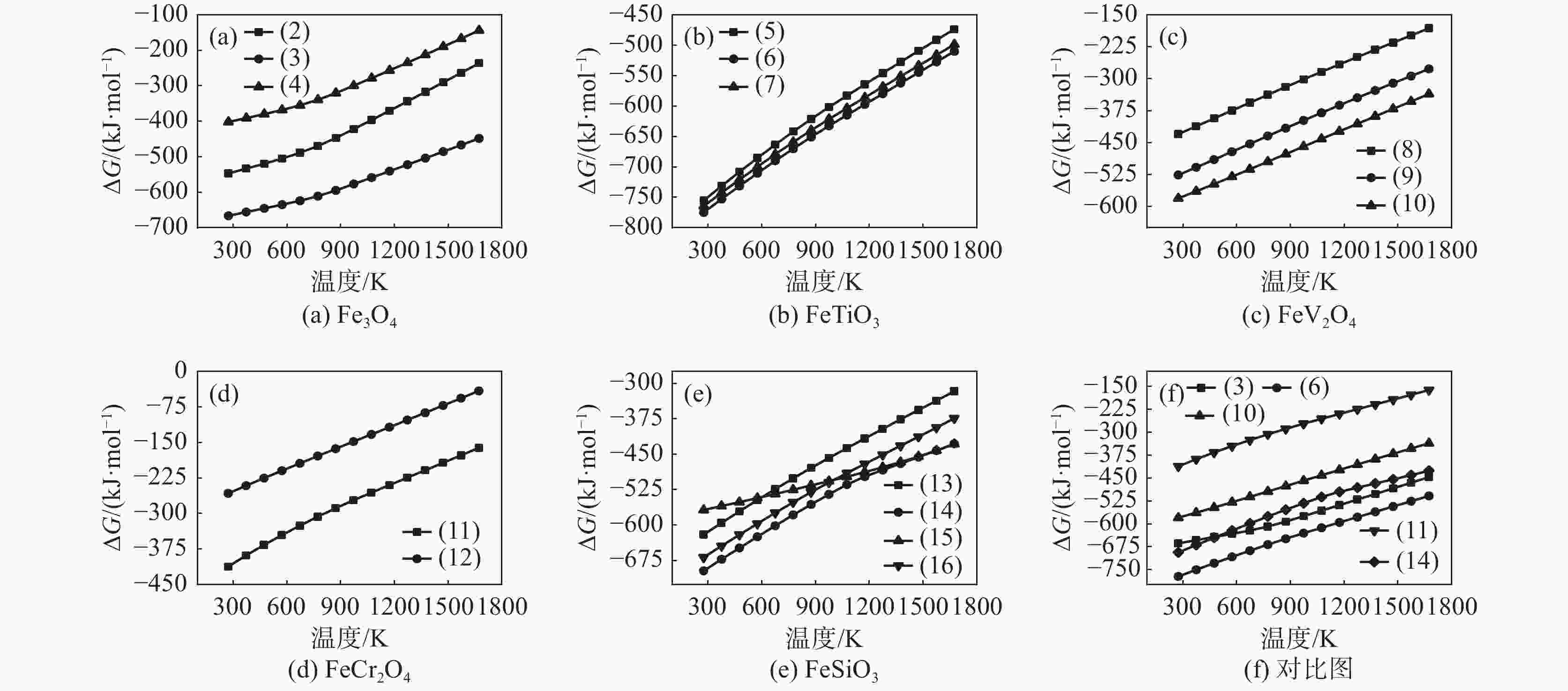
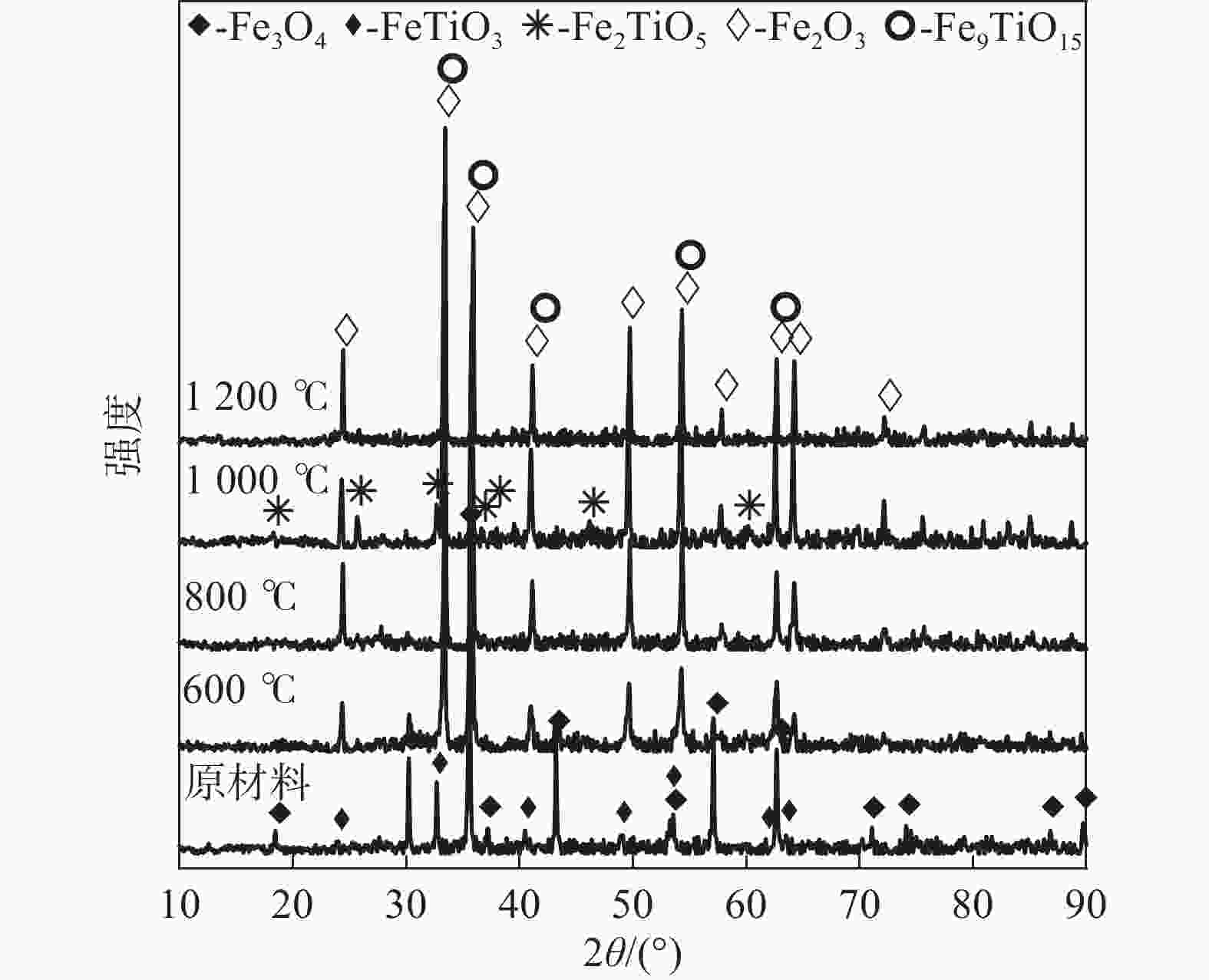
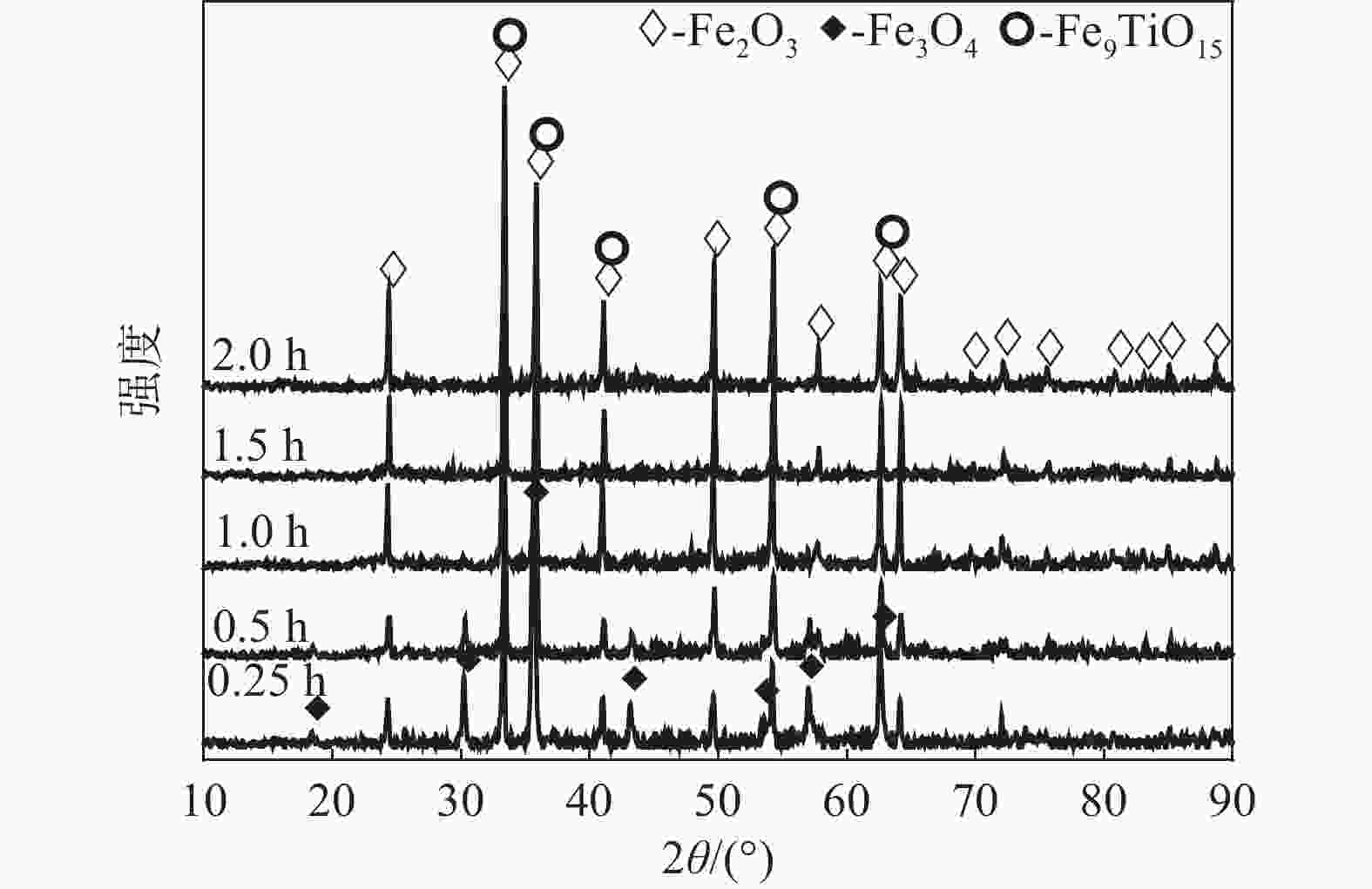
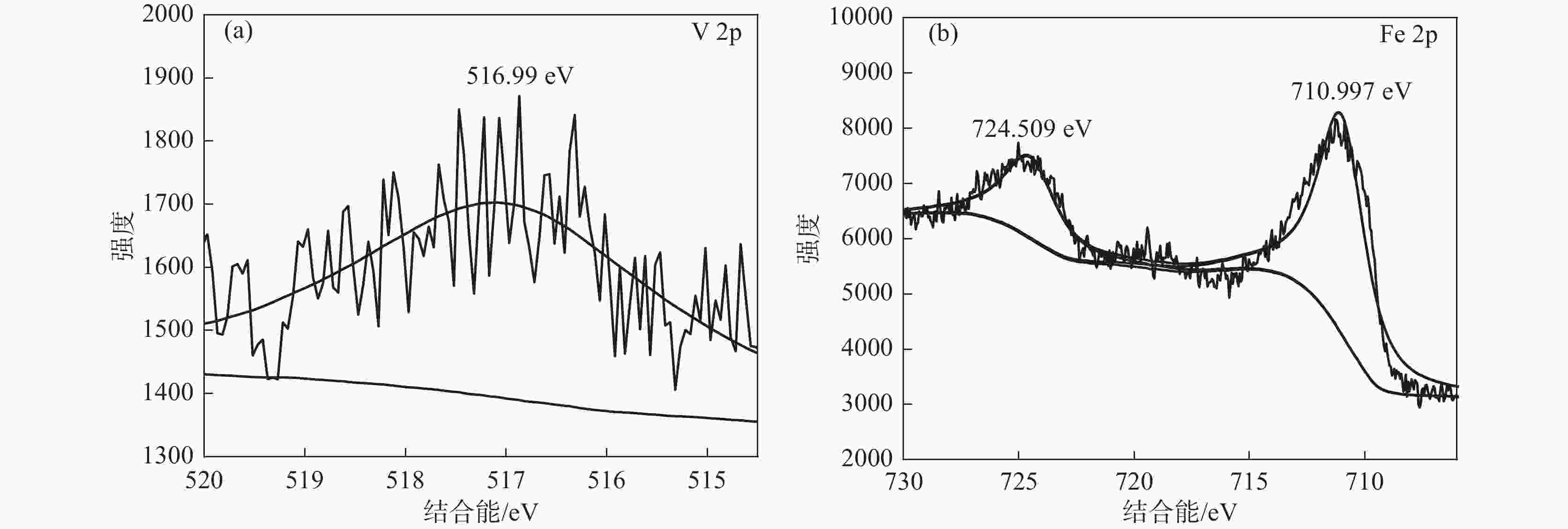
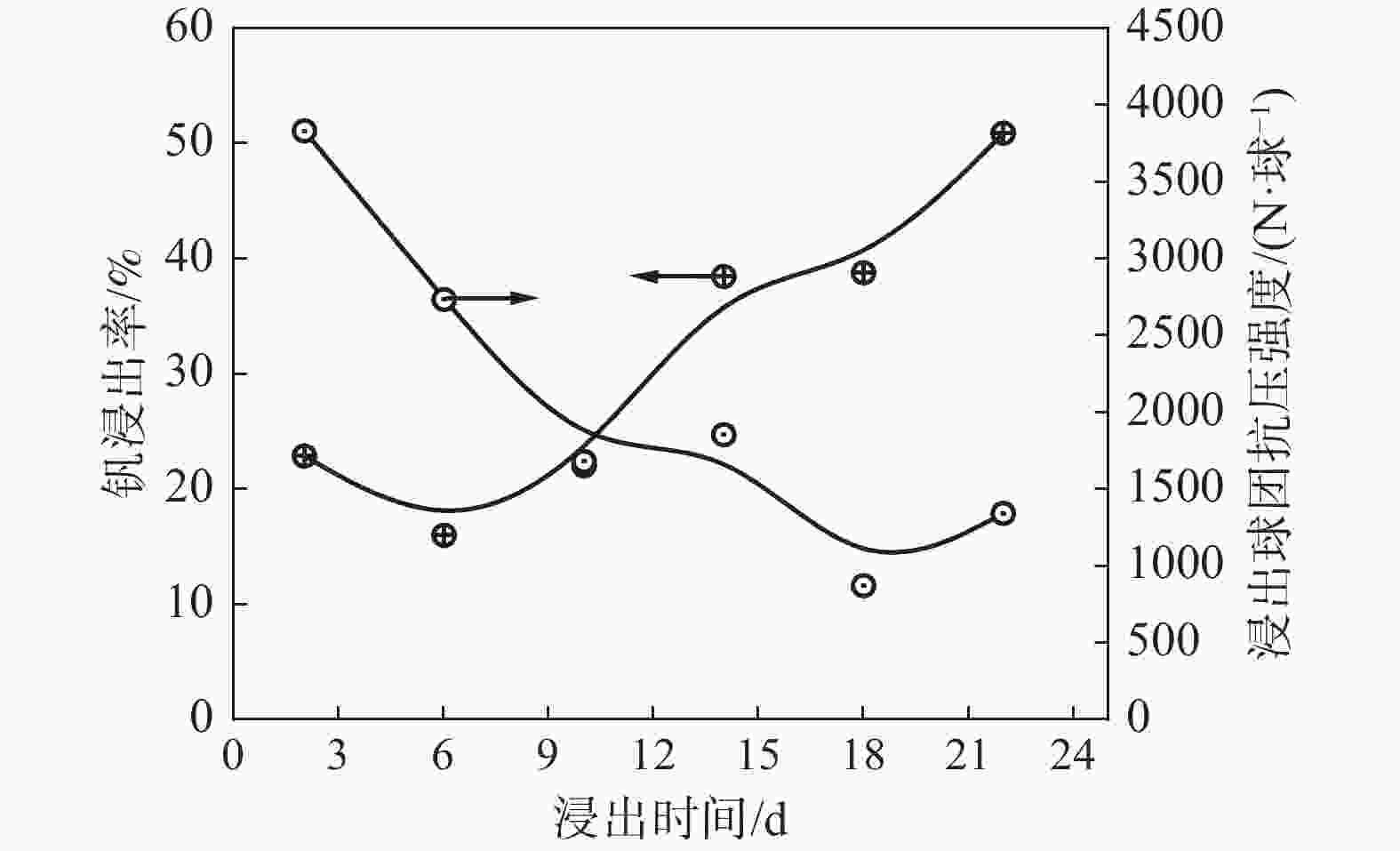
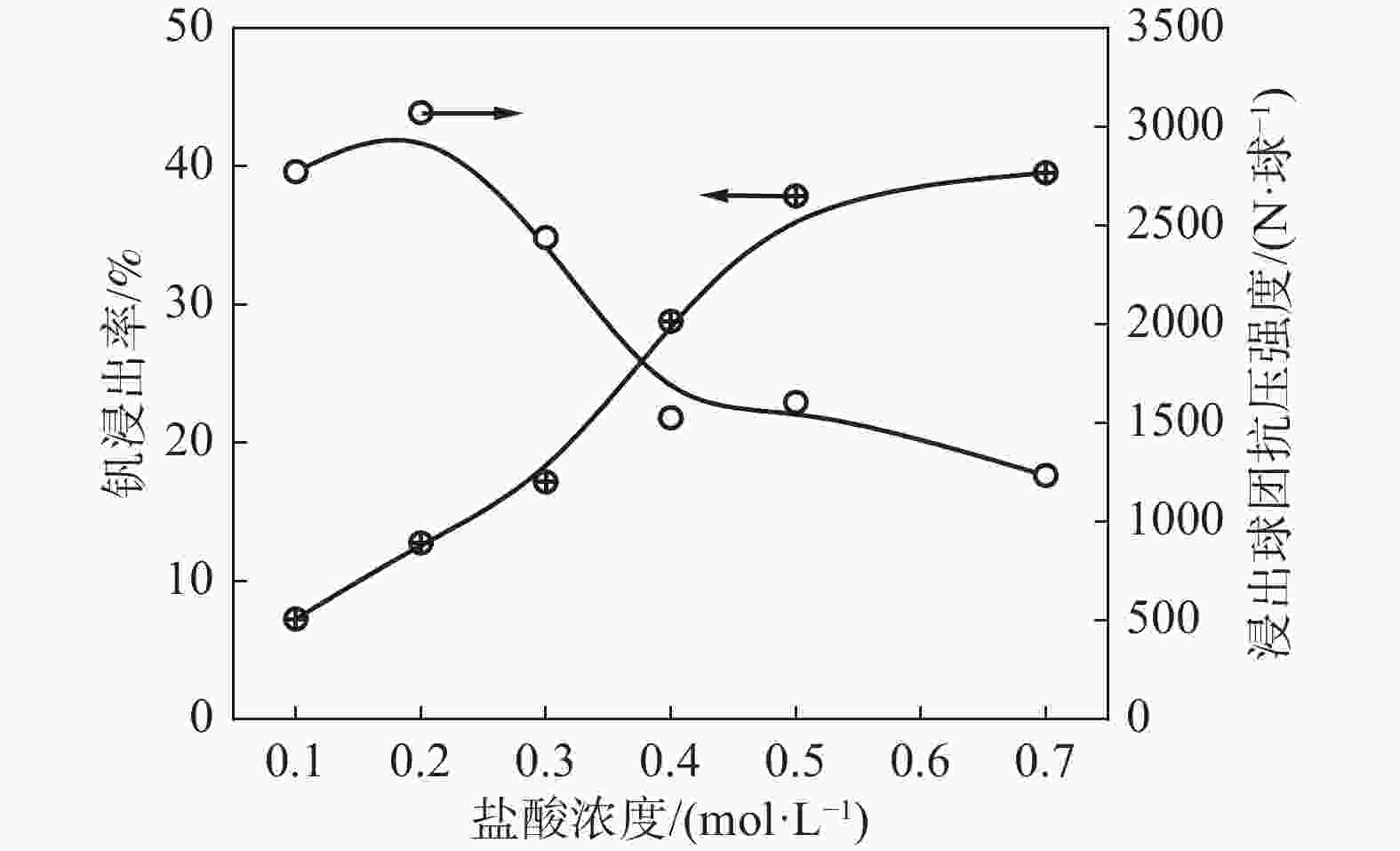
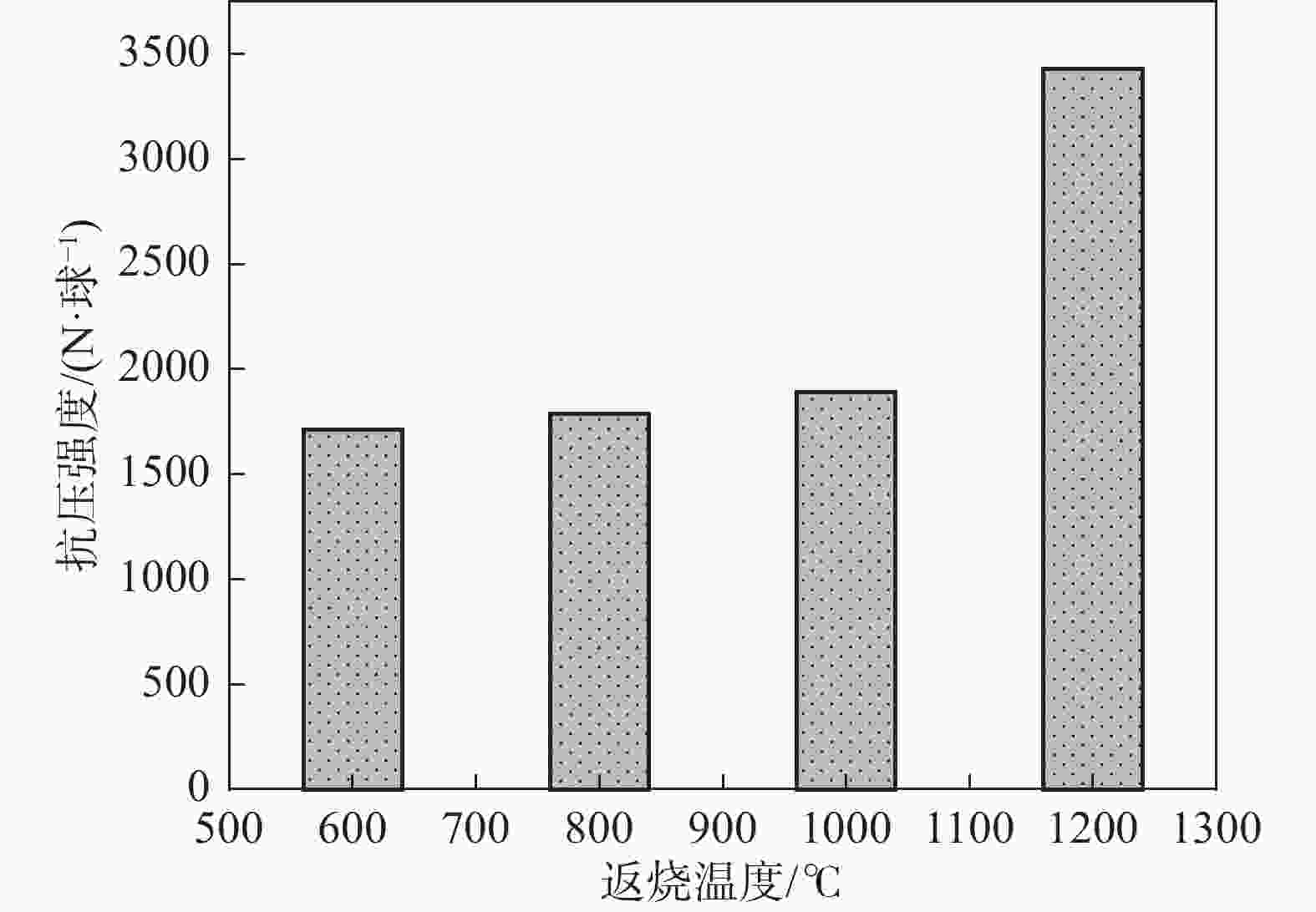
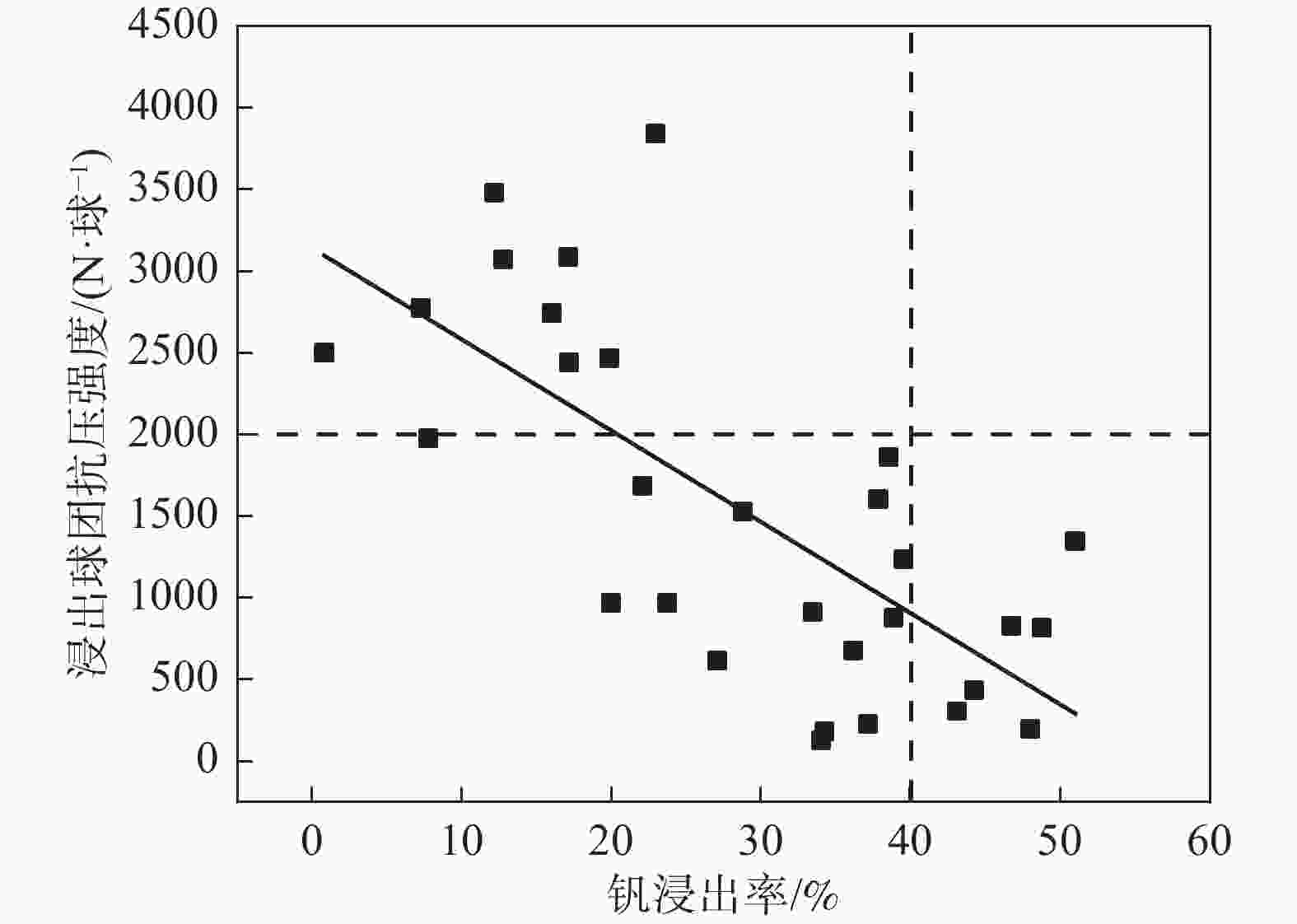
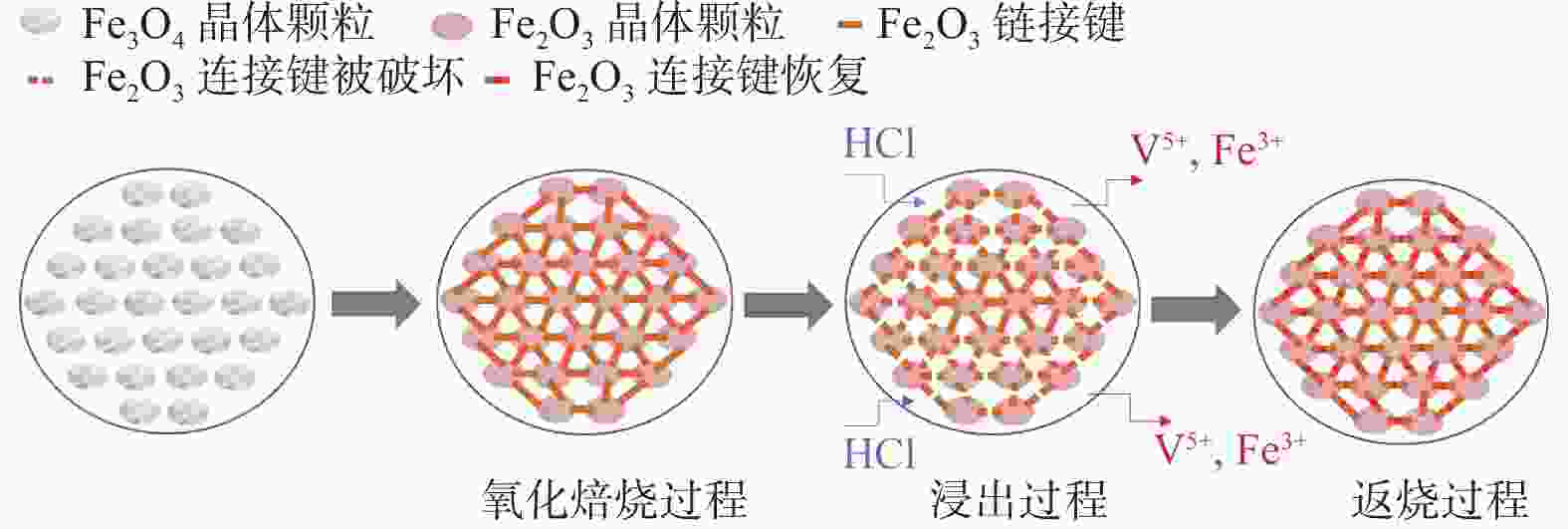
摘要: 以高铬型钒钛铁精矿为原料,研究其在氧化焙烧-盐酸浸出过程中球团强度与钒浸出行为之间的相互关系。热力学分析结果表明,在氧化钙存在的条件下,高铬型钒钛铁精矿中主要化合物的氧化反应顺序依次为:FeTiO3>Fe3O4>Fe2SiO4>FeV2O4>FeCr2O4。氧化产物物相分析结果证实,钛铁矿比磁铁矿更易氧化,氧化产物的主要物相为Fe2O3和Fe9TiO15。氧化温度和盐酸浓度分别是氧化过程和浸出过程中影响钒浸出率和浸出后球团强度的关键因素;浸出后球团强度与V2O5浸出率呈现较为明显的负相关关系;返烧可以有效提升浸出后球团强度,在返烧温度1200 ℃,返烧时间90 min条件下,返烧后球团强度大于3 000 N/球。Abstract: The relationship between crushing strength of pellets and leaching behaviors of vanadium in oxidation roasting-HCl leaching process from high-chromium vanadium-bearing titanomagnetite concentrates were investigated in this work. The results of the thermodynamic analysis of oxidizing roasting process show that the order of reaction of main compounds is: FeTiO3>Fe3O4>Fe2SiO4>FeV2O4>FeCr2O4. The phase analysis results of oxidation roasting process show that the oxidation reaction of the ilmenite is easier to magnetite, and the main phases of oxidation pellets are Fe2O3 and Fe9TiO15. Experimental results indicate that the oxidation temperature and HCl concentration are the key factors affecting the leaching efficiency of vanadium and the crushing strength of leached pellets in the oxidation process and leaching process respectively. There is an obvious negative correlation between the leaching efficiency of vanadium and the crushing strength of leached pellets. The crushing strength of leached pellets can be effectively increased by return roasting process. The crushing strength of pellets after return roasting is greater than 3 000 N/pellet under the condition of roasting temperature of 1 200 ℃ for 90 min, which can be used as raw materials for blast furnace ironmaking.
-
表 1 高铬型钒钛铁精矿的主要化学成分
Table 1. Main chemical composition of high chromium-bearing vanadium titanomagnetite concentrates
% Fe3O4 FeO TiO2 V2O5 Cr2O3 MgO Al2O3 SiO2 CaO SO3 MnO 58.61 9.93 11.03 0.69 0.67 7.57 3.79 5.46 0.69 1.02 0.25 表 2 高铬型钒钛铁精矿的粒度组成
Table 2. Size distribution of high chromium-bearing vanadium titanomagnetite concentrates
粒度/μm 占比/% <45 14.9 45~75 53.8 75~125 23.2 >125 8.1 表 3 氧化焙烧条件对氧化球团强度、浸出球团强度和钒浸出的影响正交试验
Table 3. Orthogonal test on effect of oxidizing roasting conditions on CS of oxidized pellets, CS of leached pellets and leaching efficiency of V2O5
序号 氧化温度/
℃氧化时间/
hCa(OH)2添加量/
%膨润土添加量/
%氧化球团抗压
强度/(N.球−1)浸出球团抗压
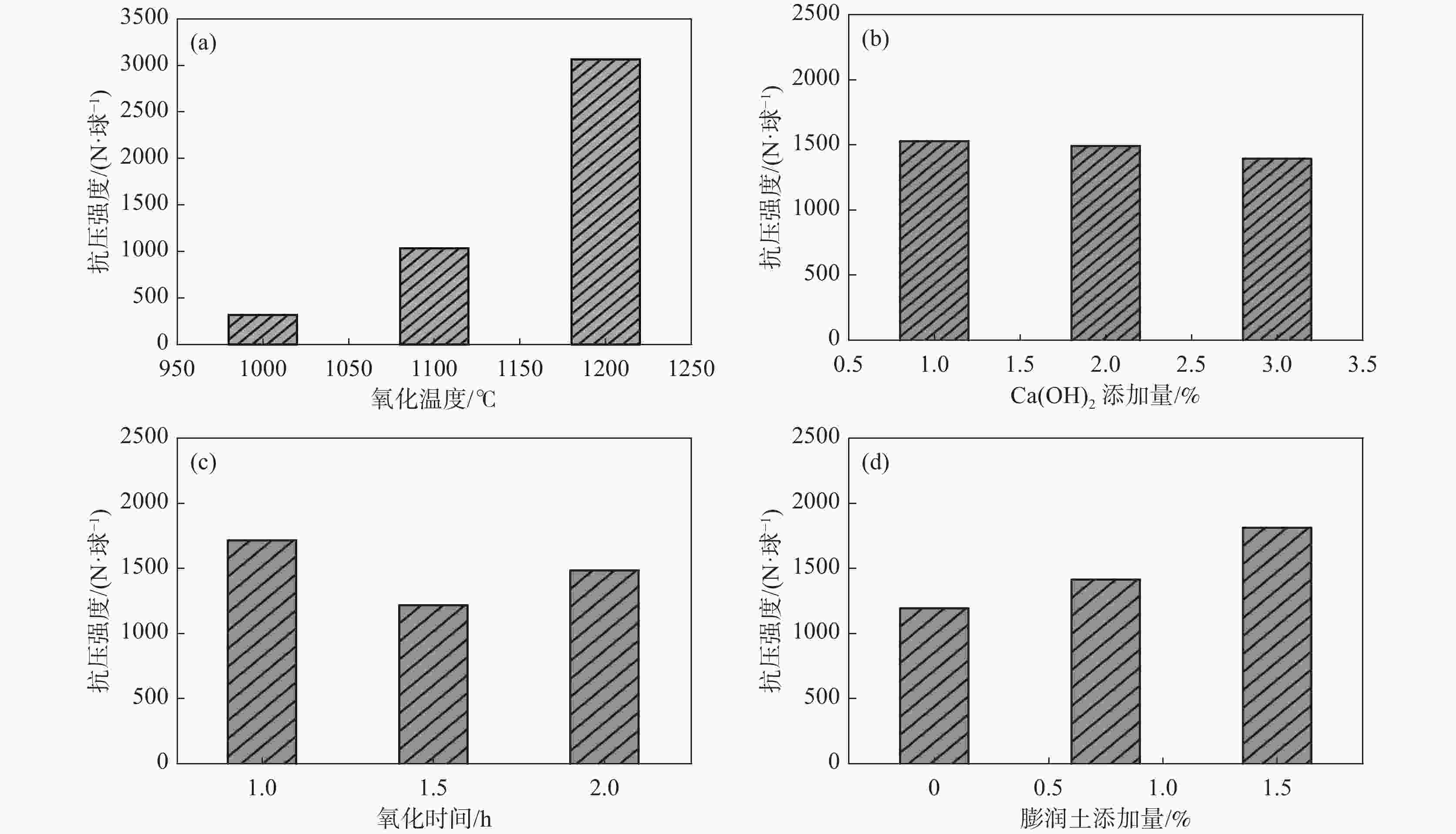
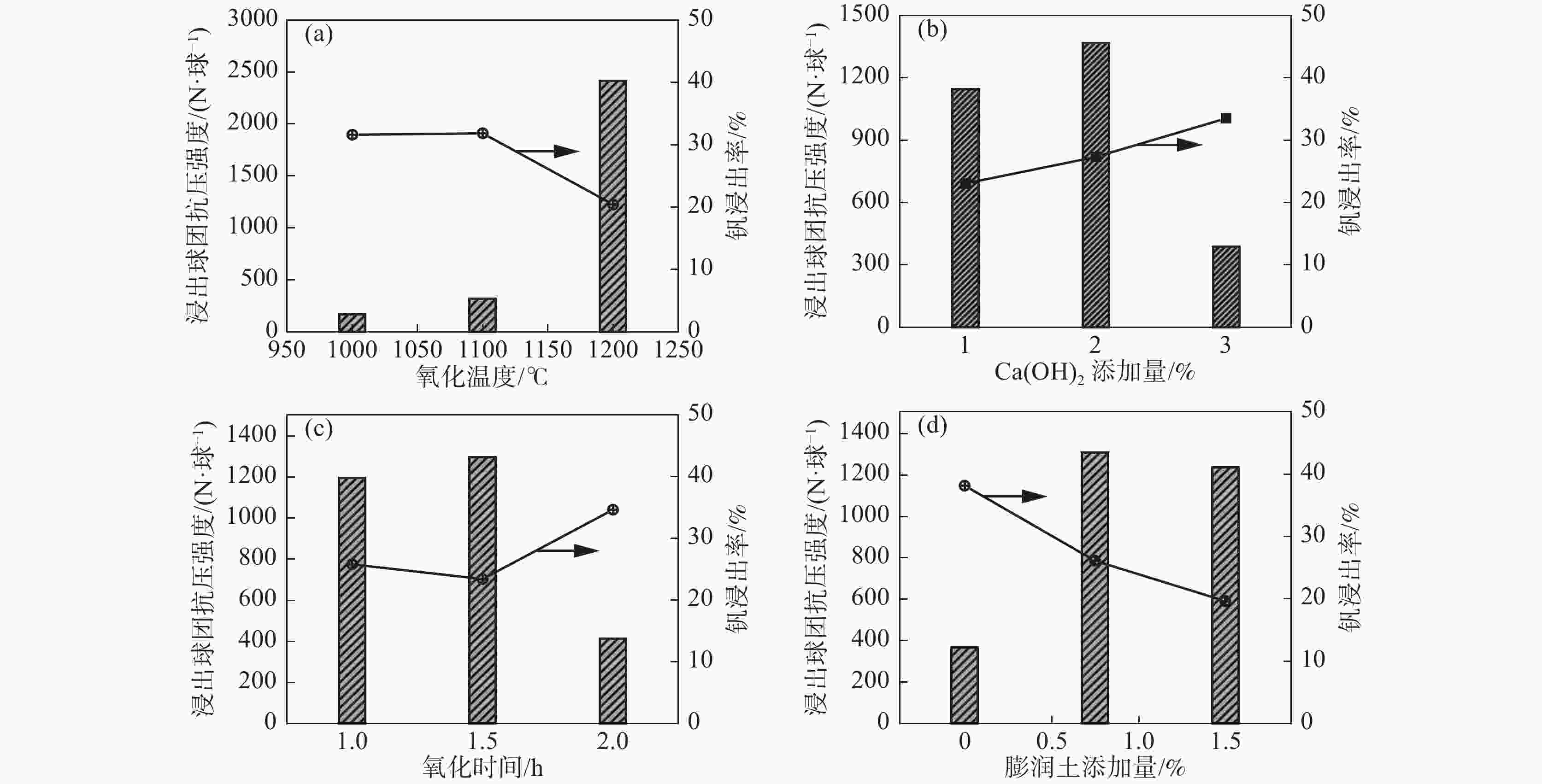
强度/(N.球−1)V2O5浸出率/% 1 1200 1 1 1.5 3707.7 3084.1 17.13 2 1200 2 1.5 0.75 2772.8 3477.0 12.19 3 1200 3 2 0 2720.5 677.2 36.17 4 1100 1 1.5 0 555.8 225.6 37.15 5 1100 2 2 1.5 1404.7 431.1 44.24 6 1100 3 1 0.75 1140.2 305.3 43.12 7 1000 1 2 0.75 325.6 127.2 34.00 8 1000 2 1 0 300.4 195.8 48.00 9 1000 3 1.5 1.5 322.8 183.0 34.26 氧化球团
抗压强度K1 3067.0 1529.7 1716.1 1811.7 K2 1033.6 1492.6 1217.1 1412.9 K3 316.3 1394.5 1483.6 1192.2 极差 R 2750.7 135.2 499.0 619.5 浸出球团
抗压强度K1 2412.8 1145.6 1195.1 1232.7 K2 320.7 1368.0 1295.2 1303.2 K3 168.7 388.5 411.8 366.2 极差 R 2244.1 979.5 883.4 937.0 V2O5浸出率 K1 21.83 29.43 36.09 31.88 K2 41.50 34.81 27.87 29.77 K3 38.75 37.85 38.14 40.44 极差 R 19.67 8.42 10.27 10.67 表 4 浸出条件对浸出球团强度和钒浸出的影响正交试验
Table 4. Orthogonal test on effect of leaching conditions on CS of leached pellets and leaching efficiency of V2O5
序号 浸出
温度/
℃浸出
时间/
d液固比 盐酸
浓度
/(mol·L−1)浸出球团
抗压强度/
(N·球−1)V2O5
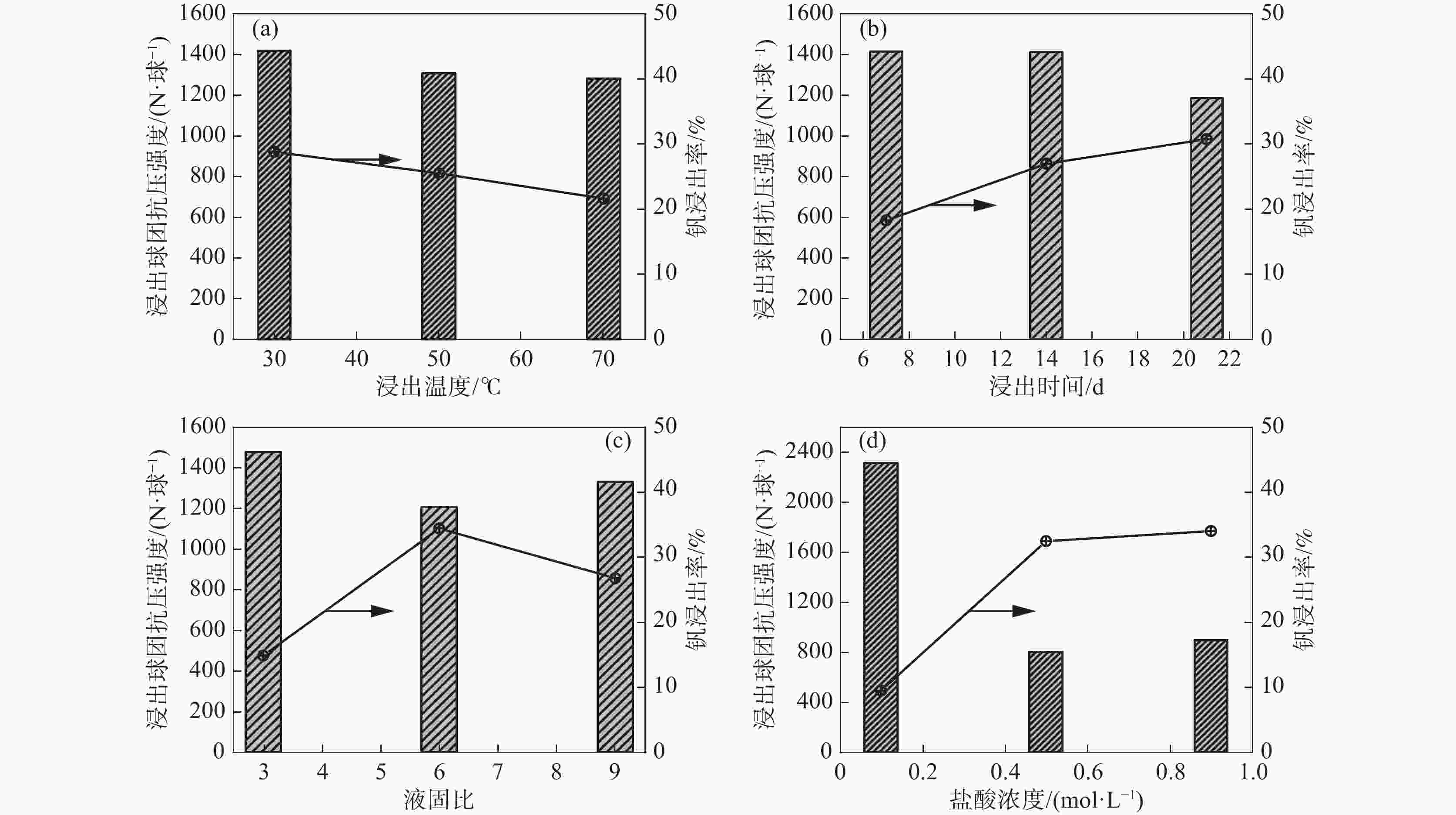
浸出率/%1 30 21 9 0.1 2466.1 19.85 2 30 14 6 0.5 828.8 46.73 3 30 7 3 0.9 966.2 19.97 4 50 21 6 0.9 815.3 48.75 5 50 14 3 0.1 2500.0 0.81 6 50 7 9 0.5 614.6 27.06 7 70 21 3 0.5 966.3 23.73 8 70 14 9 0.9 912.6 33.45 9 70 7 6 0.1 1975.7 7.78 浸出球
团抗压
强度K1 1420.4 1185.5 1331.1 2313.9 K2 1310.0 1413.8 1206.6 803.2 K3 1284.9 1415.9 1477.5 898.0 极差 R 135.5 230.4 270.9 1510.7 V2O5
浸出率K1 28.85 30.78 26.78 9.48 K2 25.54 26.99 34.42 32.50 K3 21.65 18.27 14.84 34.06 极差 R 7.20 12.51 19.58 24.58 -
[1] Gao Shimin. Discussion on smelting technology of a high chromium vanadium titanium magnetite[J]. Iron Steel Vanadium Titanium, 2020,41(5):27−36. (高师敏. 一种高铬型钒钛磁铁矿冶炼工艺探讨[J]. 钢铁钒钛, 2020,41(5):27−36. doi: 10.7513/j.issn.1004-7638.2020.05.005 [2] Zhou Mi, Jing Tao, Yang Songtao, et al. Sintering behaviors and consolidation mechanism of high-chromium vanadium and titanium magnetite fines[J]. International Journal of Minerals, Metallurgy and Materials, 2015,22(9):917. doi: 10.1007/s12613-015-1150-7 [3] He Zhanwei, Xue Xiangxin. Basic sintering characteristics of several typical vanadium titanium magnetite[J]. Iron and Steel, 2020,55(5):20−25. (何占伟, 薛向欣. 典型钒钛磁铁矿的烧结基础特性[J]. 钢铁, 2020,55(5):20−25. doi: 10.13228/j.boyuan.issn0449-749x.20190410 [4] Fu Xiaojiao, Zhao Jiaqi, Chen Shuangyin, et al. Application of boron-bearing iron concentrate to optimizing sintering quality of high chromium vanadium-titanium magnetite ore[J]. Journal of Northeastern University( Natural Science), 2015,36(8):1115−1119. (付小佼, 赵嘉琦, 陈双印, 等. 硼铁矿应用于高铬型钒钛矿烧结优化的试验研究[J]. 东北大学学报(自然科学版), 2015,36(8):1115−1119. doi: 10.3969/j.issn.1005-3026.2015.08.012 [5] Zhang Liheng, Gao Zixian, Tang Weidong, et al. Effect of TiO2 content on metallurgy performance of high-chromium vanadium-titanium magnetite sinter[J]. Journal of Northeastern University( Natural Science), 2020,41(11):1667−1672. (张立恒, 高子先, 汤卫东, 等. w(TiO2)对高铬型钒钛磁铁矿烧结矿冶金性能的影响[J]. 东北大学学报(自然科学版), 2020,41(11):1667−1672. doi: 10.12068/j.issn.1005-3026.2020.11.023 [6] Liu Zhenggen, Chu Mansheng, Wang Hongtao, et al. Effect of MgO content in sinter on the softening–melting behavior of mixed burden made from chromium-bearing vanadium–titanium magnetite[J]. International Journal of Minerals, Metallurgy and Materials, 2016,23(1):25. doi: 10.1007/s12613-016-1207-2 [7] Tang Jue, Zhang Yong, Chu Mansheng, et al. Effect of the increasing percent of high chromium vanadium-titanium magnetite on quality of oxidized pellets[J]. Journal of Northeastern University( Natural Science), 2013,34(7):956−960. (唐珏, 张勇, 储满生, 等. 高铬型钒钛磁铁矿配量增加对氧化球团质量的影响[J]. 东北大学学报(自然科学版), 2013,34(7):956−960. doi: 10.3969/j.issn.1005-3026.2013.07.011 [8] Li Wei, Wang Nan, Fu Guiqin, et al. Effect of Cr2O3 addition on the oxidation induration mechanism of Hongge vanadium titanomagnetite pellets[J]. International Journal of Minerals, Metallurgy and Materials, 2018,25(4):391. doi: 10.1007/s12613-018-1583-x [9] Li Wei, Fu Guiqin, Chu Mansheng, et al. Investigation of the oxidation induration mechanism of Hongge vanadium titanomagnetite pellets with different Al2O3 additions[J]. Powder Technology, 2020,36:555−561. [10] Li Wei, Wang Nan, Fu Guiqin, et al. Influence of TiO2 addition on the oxidation induration and reduction behavior of Hongge vanadium titanomagnetite pellets with simulated shaft furnace gases[J]. Powder Technology, 2018,326:137−145. doi: 10.1016/j.powtec.2017.12.050 [11] Tang Weidong, Yang Songtao, Xue Xiangxin. Effect of B2O3 addition on oxidation induration and reduction swelling behavior of chromium-bearing vanadium titanomagnetite pellets with simulated coke oven gas[J]. Transactions of Nonferrous Metals Society of China, 2019,29(7):1549−1559. doi: 10.1016/S1003-6326(19)65062-4 [12] Tang Weidong, Yang Songtao, Xue Xiangxin. Effect of Cr2O3 addition on oxidation induration and reduction swelling behavior of chromium-bearing vanadium titanomagnetite pellets with simulated coke oven gas[J]. International Journal of Minerals, Metallurgy and Materials, 2019,26(8):963. doi: 10.1007/s12613-019-1813-x [13] Zhou Shifa, Zheng Haiyan, Dong Yue. Reduction dynamics of carbon-containing pellets of vanadium-bearing titanomagnetite[J]. Iron and Steel, 2021,56(6):15−20. (周诗发, 郑海燕, 董越, 等. 钒钛磁铁矿含碳球团的还原历程[J]. 钢铁, 2021,56(6):15−20. doi: 10.13228/j.boyuan.issn0449-749x.20200510 [14] Zhao Longsheng, Wang Lina, Chen Desheng, et al. Behaviors of vanadium and chromium in coal-based direct reduction of high-chromium vanadium-bearing titanomagnetite concentrates followed by magnetic separation[J]. Transactions of Nonferrous Metals Society of China, 2015,25(4):1325−1333. doi: 10.1016/S1003-6326(15)63731-1 [15] Yang Songtao, Zhou Mi, Jiang Tao, et al. Application of a water cooling treatment and its effect on coal-based reduction of high-chromium vanadium and titanium iron ore[J]. International Journal of Minerals, Metallurgy and Materials, 2016,23(12):1353. doi: 10.1007/s12613-016-1358-1 [16] Tang Jue, Chu Mansheng, Li Feng, et al. Reduction mechanism of high-chromium vanadium–titanium magnetite pellets by H2-CO-CO2 gas mixtures[J]. International Journal of Minerals, Metallurgy and Materials, 2015,22(6):562. doi: 10.1007/s12613-015-1108-9 [17] Li Wei, Fu Guiqin, Chu Mansheng, et al. Reduction behavior and mechanism of Hongge vanadium titanomagnetite pellets by gas mixture of H2 and CO[J]. Journal of Iron and Steel Research, International, 2017,24(1):34−42. doi: 10.1016/S1006-706X(17)30006-7 [18] Li Wei, Fu Guiqin, Chu Mansheng, et al. Effect of porosity of Hongge vanadium titanomagnetite-oxidized pellet on its reduction swelling behavior and mechanism with hydrogen-rich gases[J]. Powder Technology, 2019,343:194−203. doi: 10.1016/j.powtec.2018.11.027 [19] Feng Cong, Chu Mansheng, Tang Jue, et al. Effects of smelting parameters on the slag/metal separation behaviors of Hongge vanadium-bearing titanomagnetite metallized pellets obtained from the gas-based direct reduction process[J]. International Journal of Minerals, Metallurgy and Materials, 2018,25(6):609. doi: 10.1007/s12613-018-1608-5 [20] Zhao Longsheng, Wang Lina, Qi Tao, et al. A novel method to extract iron, titanium, vanadium, and chromium from high-chromium vanadium-bearing titanomagnetite concentrates[J]. Hydrometallurgy, 2014,149:106−109. doi: 10.1016/j.hydromet.2014.07.014 [21] Luo Yi, Che Xiaokui, Cui Xinglan, et al. Selective leaching of vanadium from V-Ti magnetite concentrates by pellet calcification roasting-H2SO4 leaching process[J]. International Journal of Mining Science and Technology, 2021,31(3):507−513. doi: 10.1016/j.ijmst.2021.02.002 [22] Yamasgita Toru, Hayes Peter. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials[J]. Applied Surface Science, 2008,254(8):2441−2449. doi: 10.1016/j.apsusc.2007.09.063 [23] 吴祥龙. 高铬型钒钛磁铁矿氧化焙烧-气基直接还原过程机理实验研究[D]. 沈阳: 东北大学, 2013.Wu Xianglong. Experiment study on the process mechanism of oxidizing roasting and gas-based direct reduction of high chromium vanadium-titanium magnetite[D]. Shenyang: Northeastern University, 2013. -





 下载:
下载: