Effect of Sc3+ doping on lithium storage performance of Li3V2(PO4)3/C cathode material synthesized by carbothermal reduction method
-
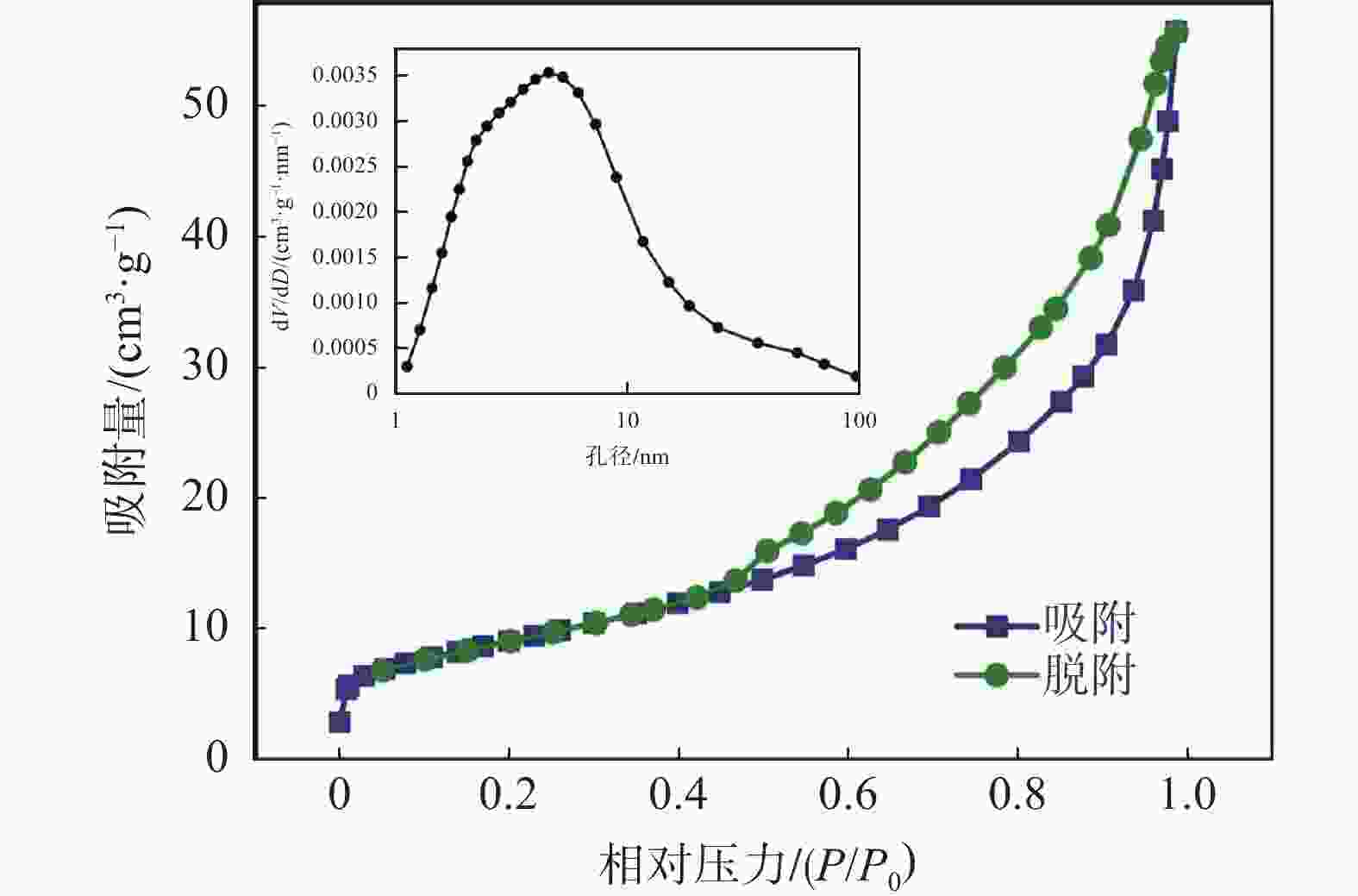
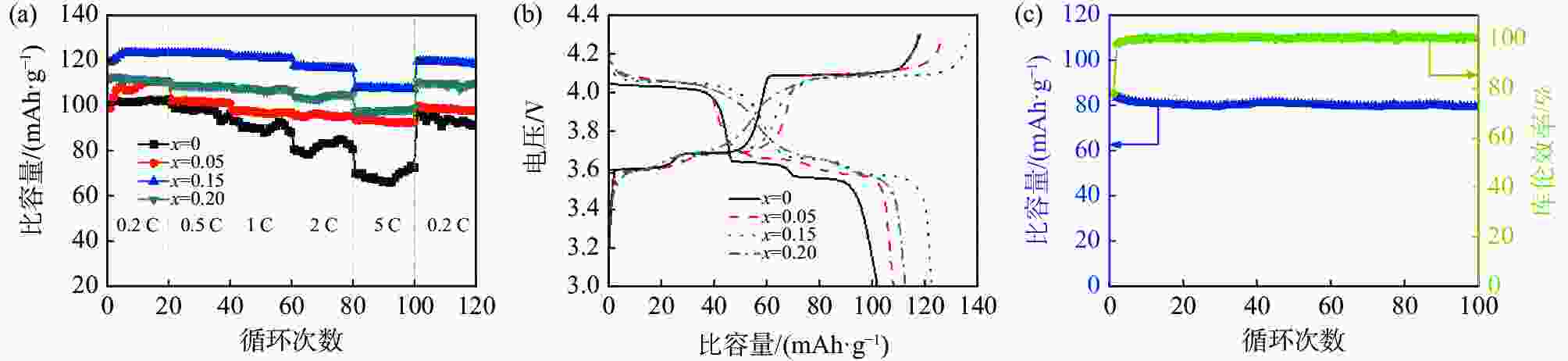
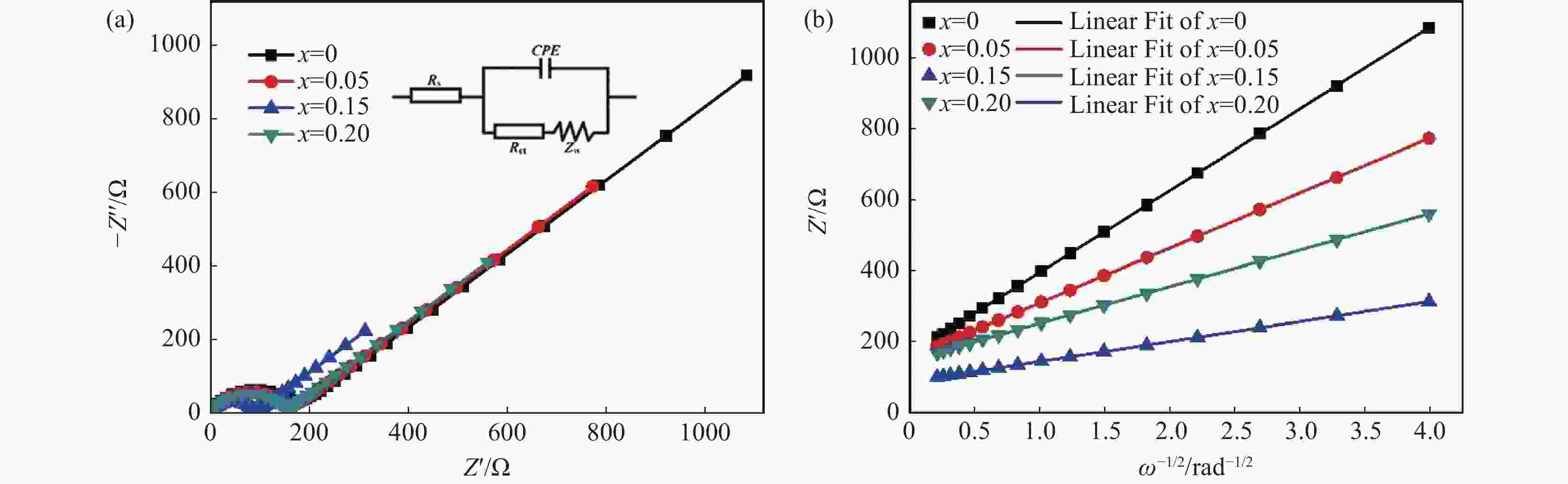
摘要: 通过碳热还原法成功制备出Sc3+掺杂Li3V2(PO4)3/C正极材料。系统研究了Sc3+掺杂量对Li3V2(PO4)3的结构、形貌及电化学性能的影响。Sc3+掺杂虽然没有改变Li3V2(PO4)3的晶格类型,但是使得Li3V2(PO4)3的晶格膨胀,晶胞体积增大,有利于电子传输和Li+扩散。此外,Sc3+掺杂使得不规则的多边形块状Li3V2(PO4)3颗粒球化并减小其尺寸。更为重要的是,合适的Sc3+掺杂量能显著增强Li3V2(PO4)3正极材料的电子电导率和Li+扩散系数。得益于适当的Sc3+掺杂量以及碳包覆和多孔结构,Li3V1.85Sc0.15(PO4)3/C样品具有优异的储锂性能,其在10 C的高倍率下可提供84.8 mAh/g的首次放电比容量,并且循环100圈后容量保持率高达93.5%。
-
关键词:
- Li3V2(PO4)3 /
- Sc3+掺杂 /
- 碳热还原法 /
- 储锂性能 /
- 放电比容量
Abstract: In this paper, Sc3+ doped Li3V2(PO4)3/C cathode material was successfully prepared by carbothermal reduction method. The effects of Sc3+ doping amount on the structure, morphology and lithium storage properties of Li3V2(PO4)3 were systematically investigated. Although Sc3+ doping does not change the lattice type of Li3V2(PO4)3, it makes the lattice of Li3V2(PO4)3 expand and the unit cell volume increase, which is beneficial to electron transport and Li+ diffusion. In addition, Sc3+ doping makes irregular polygonal Li3V2(PO4)3 particles spheroidized and reduces the particle size. More importantly, the appropriate doping amount of Sc3+ can significantly enhance the electronic conductivity and Li+ diffusion coefficient of Li3V2(PO4)3 cathode material. Benefiting from the appropriate Sc3+ doping amount, carbon coating and porous structure, Li3V1.85Sc0.15(PO4)3/C samples possess superior lithium storage properties. The initial discharge specific capacity is 84.8 mAh/g at 10 C rate, and the capacity retention rate is as high as 93.5 % after 100 cycles. -
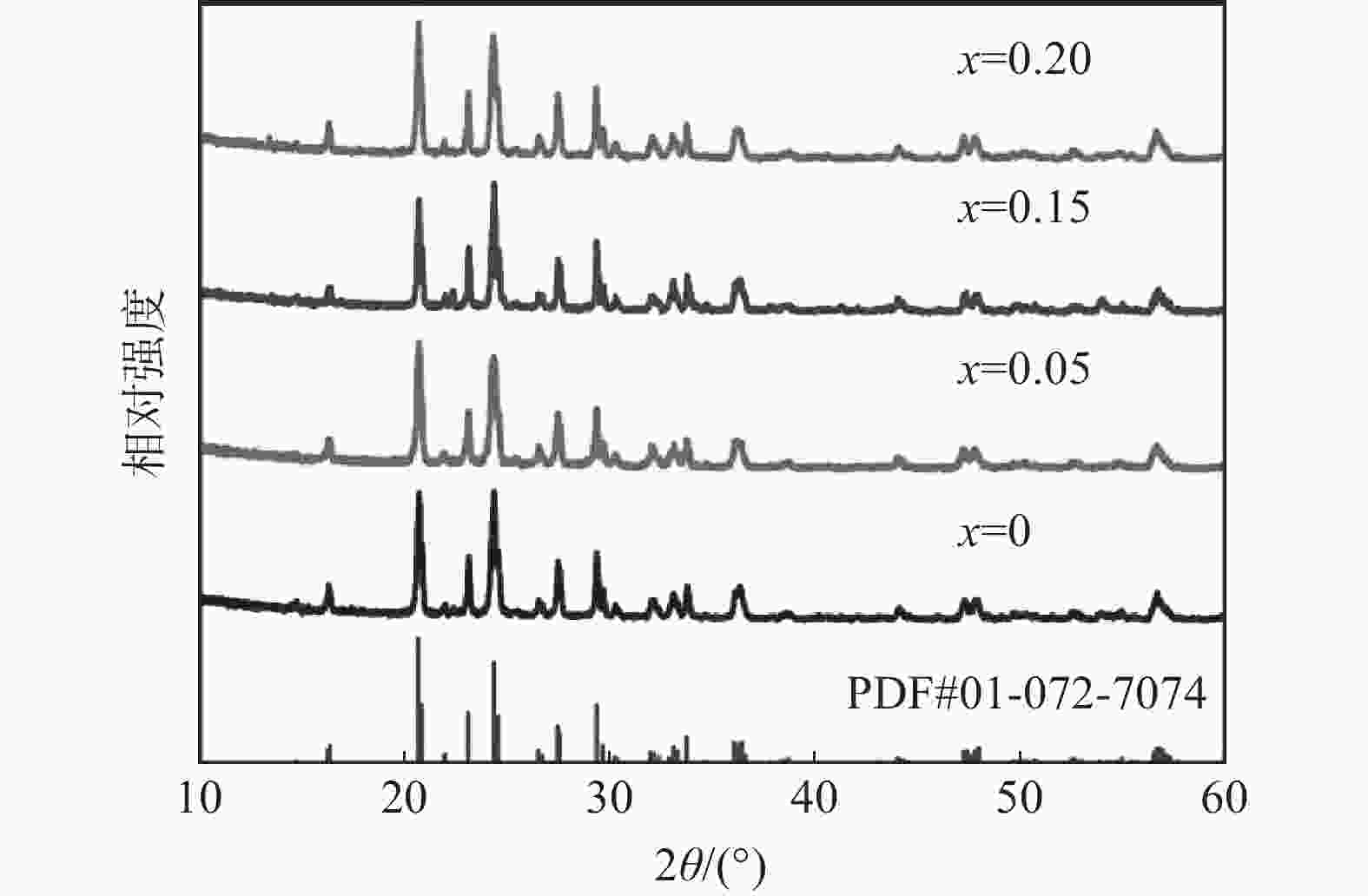
表 1 单斜Li3V2−xScx(PO4)3/C样品精修后的晶格参数和对应的晶胞体积(a,b,c和β:单斜晶系的晶胞参数;V:晶胞体积)
Table 1. Refined lattice parameters of monoclinic Li3V2−xScx(PO4)3/C materials and the corresponding unit-cell volumes (a, b, c and β : unit-cell parameters of the monoclinic system; V : volume of unit-cell)
样品 a /nm b /nm c /nm β /° V /nm3 Li3V2(PO4)3/C 0.860 1 0.859 5 1.203 90.55 0.889 6 Li3V1.90Sc0.05(PO4)3/C 0.860 8 0.860 2 1.203 90.52 0.891 0 Li3V1.85Sc0.15(PO4)3/C 0.8613 0.860 7 1.205 90.51 0.893 1 Li3V1.80Sc0.20(PO4)3/C 0.862 1 0.861 7 1.206 90.48 0.895 6 -
[1] Oh W, Park H, Jin B S, et al. Understanding the structural phase transitions in lithium vanadium phosphate cathodes for lithium-ion batteries[J]. Journal of Materials Chemistry A, 2020,8(20):10331−10336. doi: 10.1039/C9TA12435G [2] Ding Xiaokai, Zhang Lulu, Yang Xuelin, et al. Anthracite-derived dual-phase carbon-coated Li3V2(PO4)3 as high-performance cathode material for lithium ion batteries[J]. ACS Applied Materials & Interfaces, 2017,9(49):42788−42796. [3] Sørensen D R, Mathiesen J K, Ravnsbæk D B. Dynamic charge-discharge phase transitions in Li3V2(PO4)3 cathodes[J]. Journal of Power Sources, 2018,396:437−443. doi: 10.1016/j.jpowsour.2018.06.023 [4] Bi Linnan, Song Zhicui, Liu Xiaoqin, et al. Critical roles of RuO2 nano-particles in enhancing cyclic and rate performance of LISICON Li3V2(PO4)3 cathode materials[J]. Journal of Alloys and Compounds, 2020,845:156271. doi: 10.1016/j.jallcom.2020.156271 [5] Xia Yang, Yu Liyue, Lu Chengwei, et al. Passion fruit-like structure endows Li3V2(PO4)3@C/CNT composite with superior cyclic stability and rate performance[J]. Journal of Alloys and Compounds, 2021,859:157806. doi: 10.1016/j.jallcom.2020.157806 [6] Yan Haiyan, Li Minhao, Fu Yuqiao, et al. Conductive polypyrrole-promoted Li3V2(PO4)3 nanocomposite for rechargeable lithium energy storage[J]. Journal of Physics and Chemistry of Solids, 2022,167:110787. doi: 10.1016/j.jpcs.2022.110787 [7] Liao Yuxing, Li Chao, Lou Xiaobing, et al. Carbon-coated Li3V2(PO4)3 derived from metal-organic framework as cathode for lithium-ion batteries with high stability[J]. Electrochimica Acta, 2018,271:608−616. doi: 10.1016/j.electacta.2018.03.100 [8] Yan Haiyan, Zhang Gai, Li Yongfei. Synthesis and characterization of advanced Li3V2(PO4)3 nanocrystals@conducting polymer PEDOT for high energy lithium-ion batteries[J]. Applied Surface Science, 2017,393:30−36. doi: 10.1016/j.apsusc.2016.09.156 [9] Huang Zan, Luo Peifang, Zheng Honghong, et al. Aluminum-doping effects on three-dimensional Li3V2(PO4)3@C/CNTs microspheres for electrochemical energy storage[J]. Ceramics International, 2022,48(13):18765−18772. doi: 10.1016/j.ceramint.2022.03.151 [10] Huang Zan, Luo Peifang, Zheng Honghong. Design of Ti4+-doped Li3V2(PO4)3/C fibers for lithium energy storage[J]. Ceramics International, 2022,48(6):8325−8330. doi: 10.1016/j.ceramint.2021.12.037 [11] Qi Ning, Ma Yangyang, Ren Bing, et al. Comparison of the La-doped and Gd-doped Li3V2(PO4)3/C via electrochemical tests and first-principle calculations for lithium-ion batteries[J]. Journal of Physics and Chemistry of Solids, 2021,150:109889. doi: 10.1016/j.jpcs.2020.109889 [12] Li Nali, Yu Yong, Tong Yanwei, et al. Sc3+-doping effects on porous Li3V2(PO4)3/C cathode with superior rate performance and cyclic stability[J]. Ceramics International, 2021,47(24):34218−34224. doi: 10.1016/j.ceramint.2021.08.331 [13] Li Nali, Tong Yanwei, Yi Dawei, et al. Effect of Zr4+ doping on the morphological features and electrochemical performance of monoclinic Li3V2(PO4)3/C cathode material synthesized by an improved sol-gel combustion technique[J]. Journal of Alloys and Compounds, 2021,868:158771. doi: 10.1016/j.jallcom.2021.158771 [14] Zhang Yu, Su Zhi, Ding Juan. Synthesis and electrochemical properties of Ge-doped Li3V2(PO4)3/C cathode materials for lithium-ion batteries[J]. Journal of Alloys and Compounds, 2017,702:427−431. doi: 10.1016/j.jallcom.2017.01.267 [15] Liu Liying, Lei Xingling, Tang Hui, et al. Influences of La doping on magnetic and electrochemical properties of Li3V2(PO4)3/C cathode materials for lithium-ion batteries[J]. Electrochimica Acta, 2015,151(0):378−385. [16] Ding Xiaokai, Liu Jing, Zhang Lulu, et al. High-performance Li3V2(PO4)3/C cathode material with a mixed morphology prepared by solvothermal assisted sol-gel process[J]. Ionics, 2019,25(5):2057−2067. doi: 10.1007/s11581-018-2701-5 [17] Peng Jianhong, Bao Haiping, Xu Xiaolong, et al. Electrochemical properties of Li3V2(PO4)3/C cathode materials synthesized via ethylene glycol-assisted solvothermal method[J]. Ionics, 2018,24(5):1277−1283. doi: 10.1007/s11581-017-2312-6 [18] Wang Liping, Bai Jianming, Gao Peng, et al. Structure tracking aided design and synthesis of Li3V2(PO4)3 nanocrystals as high-power cathodes for lithium ion batteries[J]. Chemistry of Materials, 2015,27(16):5712−5718. doi: 10.1021/acs.chemmater.5b02236 [19] Li Yushan, Wang Jin, Zhou Zhaofu, et al. Large-scale synthesis of porous Li3V2(PO4)3@C/AB hollow microspheres with interconnected channel as high performance cathodes for lithium-ion batteries[J]. Journal of Alloys and Compounds, 2019,774:879−886. doi: 10.1016/j.jallcom.2018.09.214 [20] Wu Ling, Zhong Shengkui, Lu Jiajia, et al. Li3V2(PO4)3/C microspheres with high tap density and high performance synthesized by a two-step ball milling combined with the spray drying method[J]. Materials Letters, 2014,115:60−63. doi: 10.1016/j.matlet.2013.10.040 [21] Peng Yi, Tan Rou, Ma Jianmin, et al. Electrospun Li3V2(PO4)3 nanocubes/carbon nanofibers as free-standing cathodes for high-performance lithium-ion batteries[J]. Journal of Materials Chemistry A, 2019,7(24):14681−14688. doi: 10.1039/C9TA02740H [22] Wei Sainan, Yao Jiming, Shi Bao. 1D highly porous Li3V2(PO4)3/C nanofibers as superior high-rate and ultralong cycle-life cathode material for electrochemical energy storage[J]. Solid State Ionics, 2017,305:36−42. doi: 10.1016/j.ssi.2017.04.019 [23] Shannon R D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides[J]. Acta Crystallographica Section A, 1976,32(5):751−767. doi: 10.1107/S0567739476001551 [24] Xia Yang, Zhang Wenkui, Huang Hui, et al. Synthesis and electrochemical properties of Nb-doped Li3V2(PO4)3/C cathode materials for lithium-ion batteries[J]. Materials Science and Engineering:B, 2011,176(8):633−639. doi: 10.1016/j.mseb.2011.02.006 [25] Sun Hongxia, Du Haoran, Yu Mengkang, et al. Vesicular Li3V2(PO4)3/C hollow mesoporous microspheres as an efficient cathode material for lithium-ion batteries[J]. Nano Research, 2019,12(8):1937−1942. doi: 10.1007/s12274-019-2461-1 [26] Li Ruhong, Sun Shuting, Liu Jianchao, et al. From rational construction to theoretical study: Li3V2(PO4)3 nanoplates with exposed {100} facets for achieving highly stable lithium storage[J]. Journal of Power Sources, 2019,442:227231. doi: 10.1016/j.jpowsour.2019.227231 [27] Lee H S, Ramar V, Kuppan S, et al. Key design considerations for synthesis of mesoporous α-Li3V2(PO4)3/C for high power lithium batteries[J]. Electrochimica Acta, 2021,372:137831. doi: 10.1016/j.electacta.2021.137831 [28] Ding Manling, Cheng Chen, Wei Qiulong, et al. Carbon decorated Li3V2(PO4)3 for high-rate lithium-ion batteries: Electrochemical performance and charge compensation mechanism[J]. Journal of Energy Chemistry, 2021,53:124−131. doi: 10.1016/j.jechem.2020.04.020 [29] Chen Yueqian, Chen Han, Xiao Li, et al. Preparation for honeycombed Li3V2(PO4)3/C composites via vacuum-assisted immersion method and their high-rates performance in lithium-ion batteries[J]. Vacuum, 2020,172:108926. doi: 10.1016/j.vacuum.2019.108926 -




 下载:
下载: