Research on preparation of nano sodium vanadium phosphate and its sodium storage properties
-
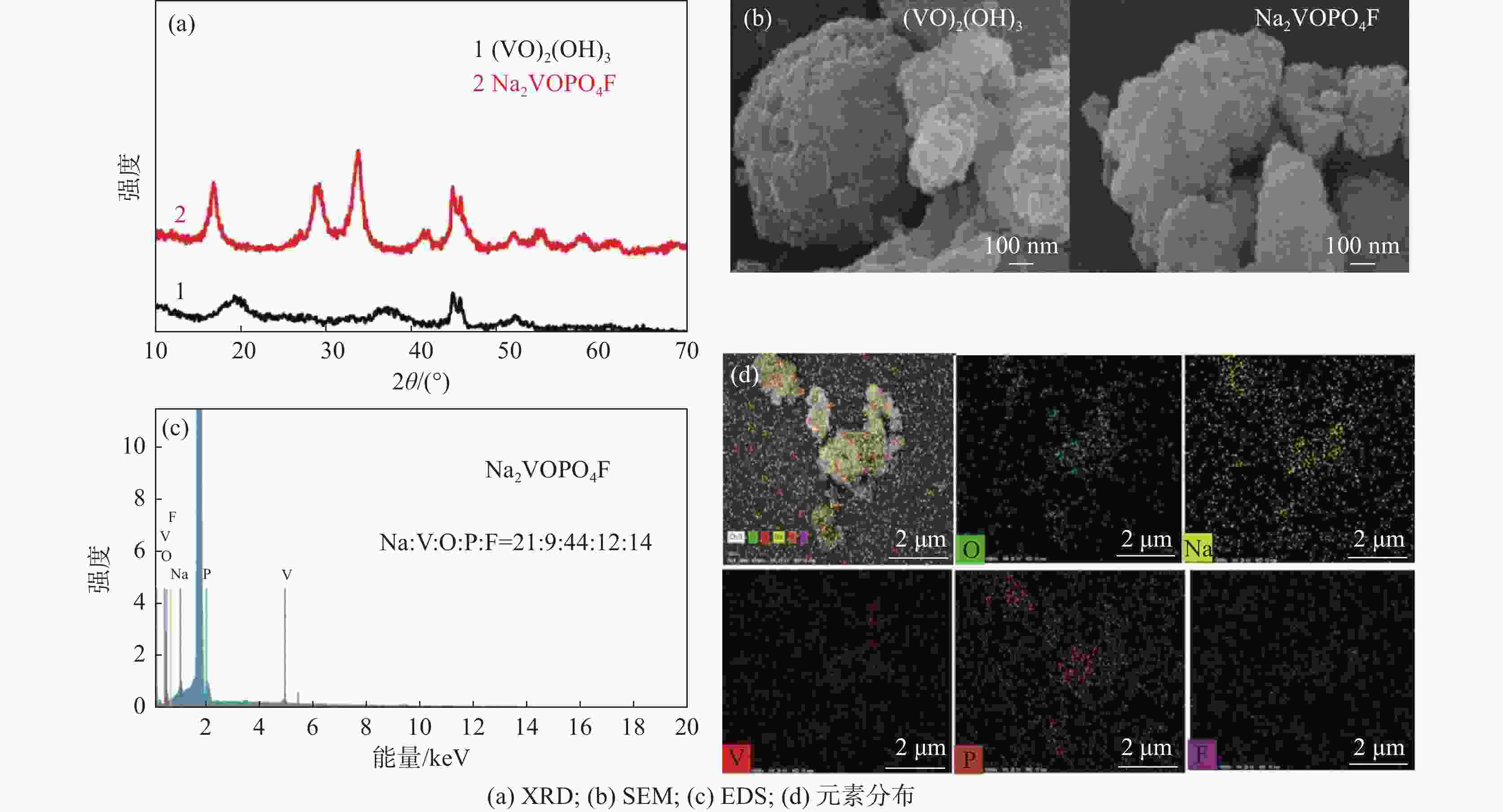
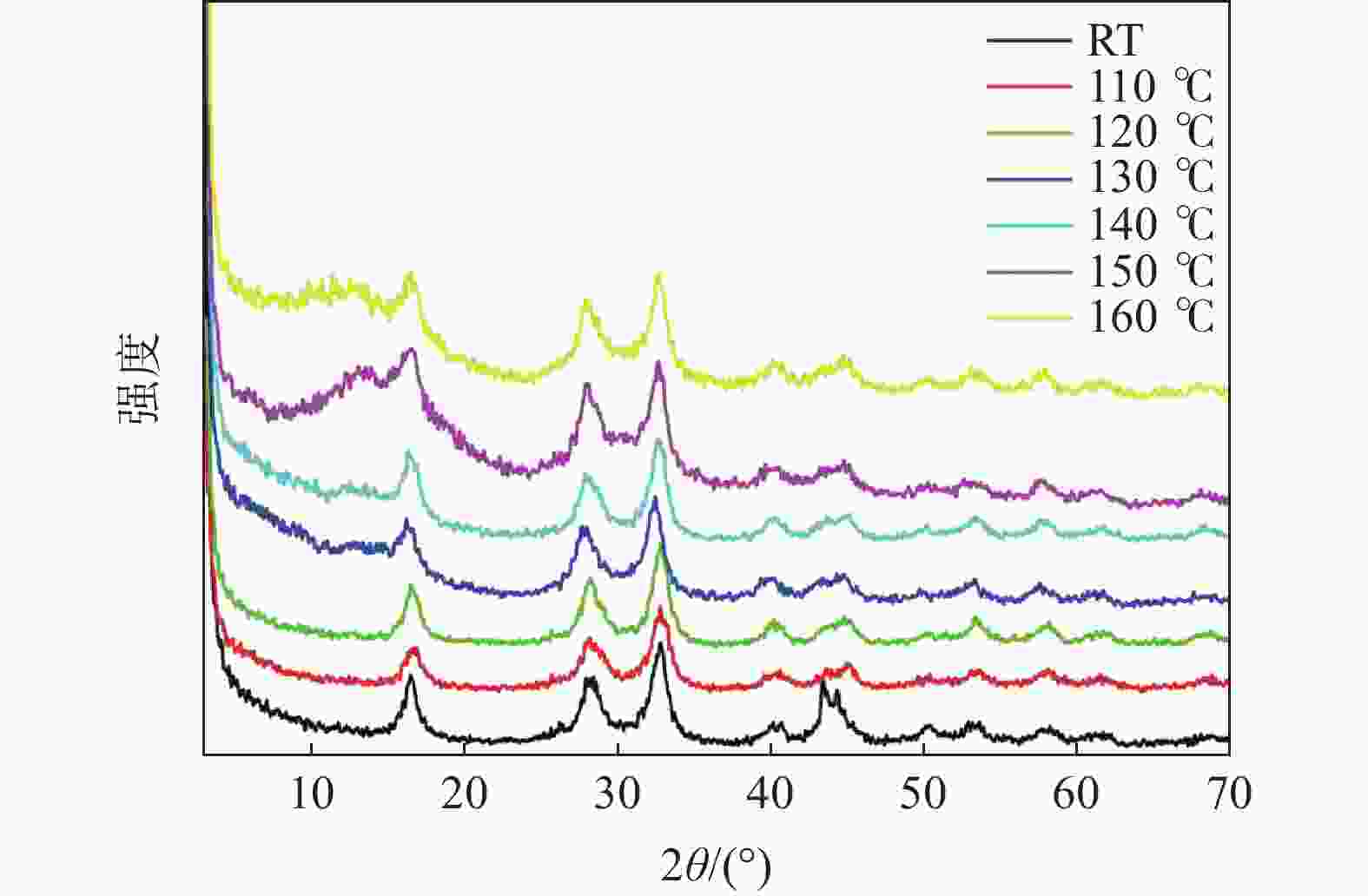
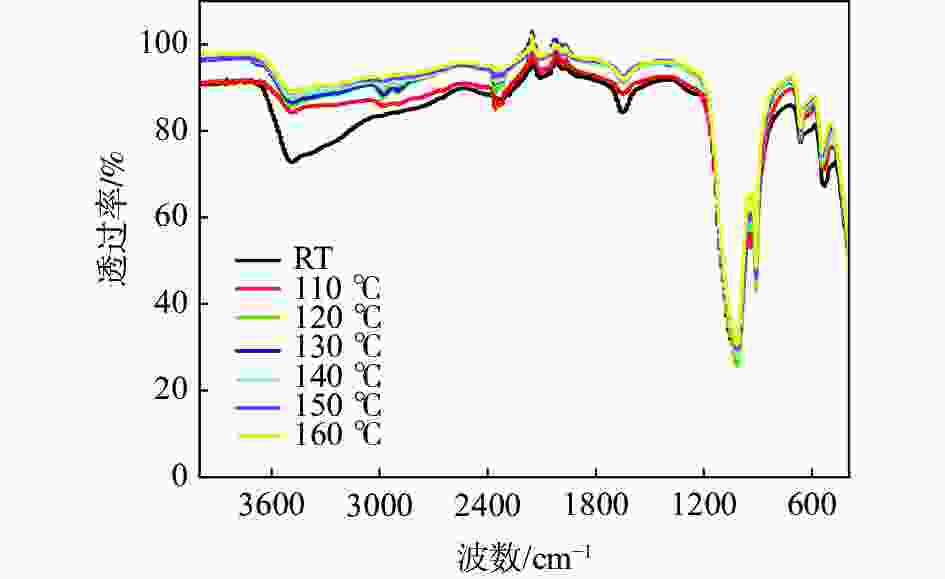
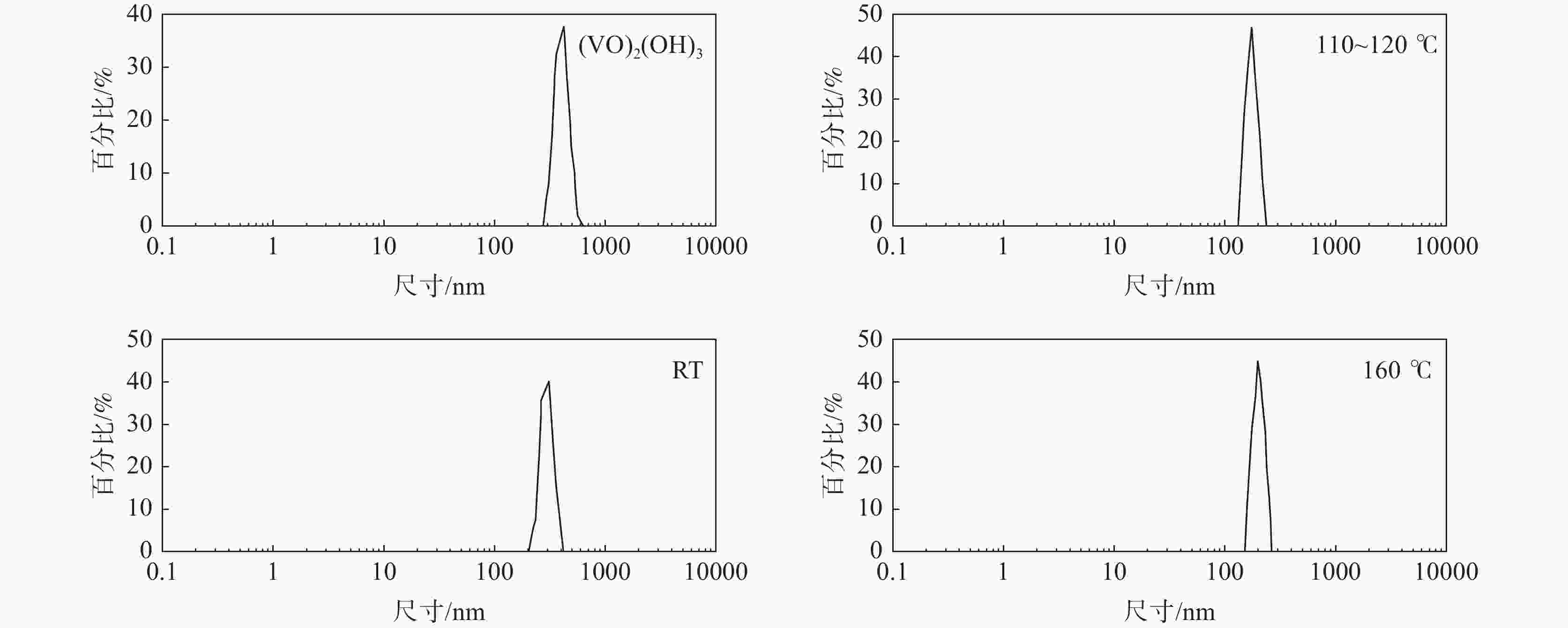
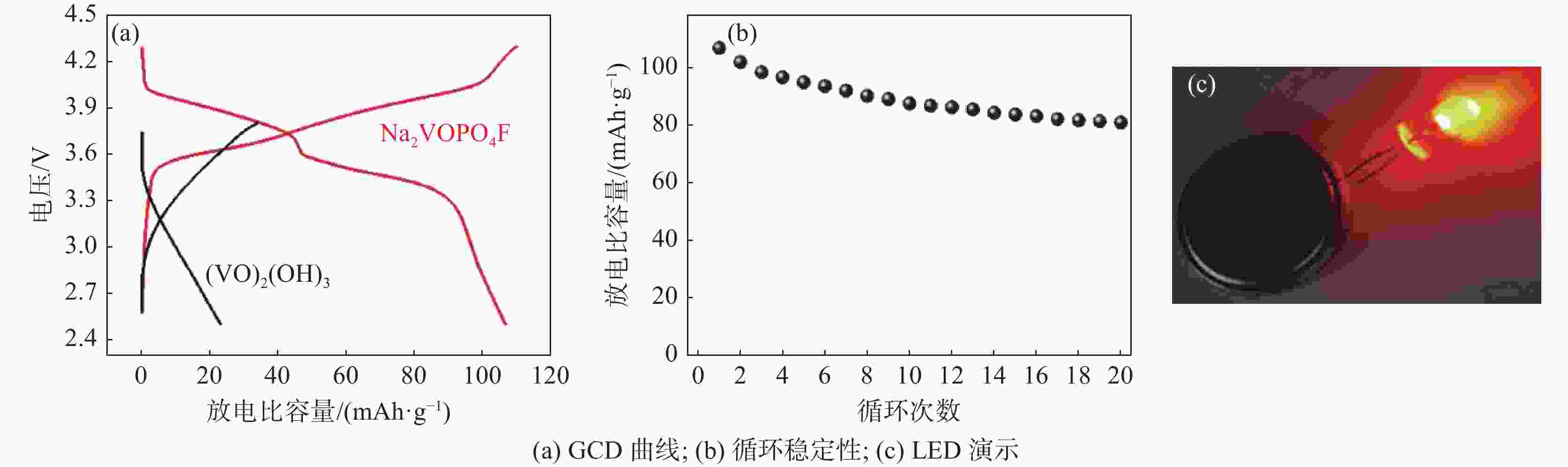
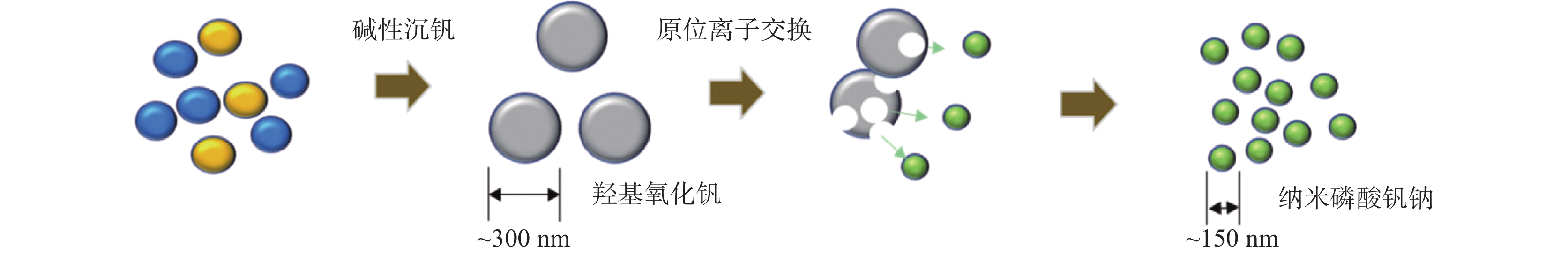
摘要: 基于磷酸钒钠制备条件苛刻、颗粒粒径大、电导率差等问题,提出了一种制备纳米磷酸钒钠的新方法。通过碱性沉钒形成羟基氧化钒,再利用PO43-、F-等阴离子与OH-的原位离子交换,从而得到纳米化的磷酸钒钠。借助XRD、SEM、FTIR等方法,分析了纳米磷酸钒钠的形成机理,优化了合成条件。电化学测试结果表明,磷酸钒钠的纳米化提升了电子/离子输运能力,使得所制备的纳米磷酸钒钠表现出优异的储钠性能。当电流密度为10 mA/g时,其放电比容量为106.68 mAh/g,并且循环20次循环充放电后,仍能保持80.85 mAh/g的放电比容量。Abstract: Based on the severe preparation conditions, large particle size and poor conductivity, a new method to prepare sodium vanadium phosphate was proposed. By the formation of alkaline vanadium compounds, vanadium hydroxyloxide, and the in-situ anions exchange reactions between PO43–, F– and OH–, nano vanadium sodium phosphate was consequently obtained. With the help of XRD, SEM, FTIR, the formation mechanism of nano vanadium sodium phosphate was analyzed, and the synthesis conditions were optimized. The electrochemical test results show that the sodium vanadium phosphate in nanoscale improves the electron/ion transport capacity, and makes the prepared sodium vanadium phosphate show excellent sodium storage performances. When the current density is 10 mA/g, the specific discharge capacity is 106.68 mAh/g. At the same time, the specific discharge capacity of 80.85 mAh/g can be maintained after 20 charge and discharge cycles.
-
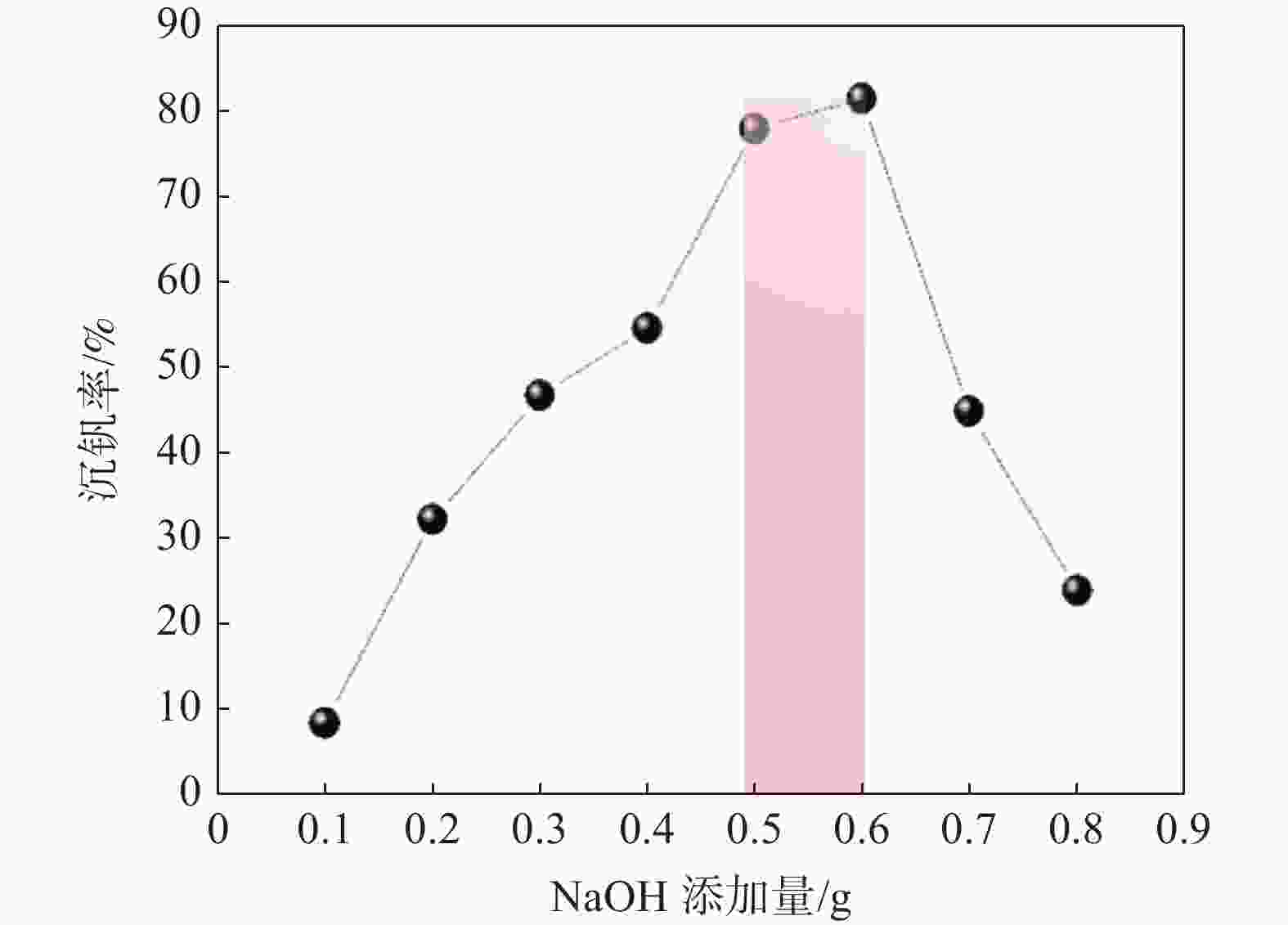
表 1 不同NaOH添加量下,羟基氧化钒的产量变化情况
Table 1. Changes in the yield of hydroxy vanadium oxide under different NaOH additions
VOSO4/
mLNaOH
添加量/gNaOH
浓度/(mol·L−1)混合
pH羟基氧化
钒产量/g沉钒
率/%10 0.1 0.25 3.36 0.0851 8.42 10 0.2 0.5 3.48 0.3245 32.13 10 0.3 0.75 3.62 0.4720 46.73 10 0.4 1 3.78 0.5514 54.59 10 0.5 1.25 3.85 0.7831 77.83 10 0.6 1.5 5.54 0.822 81.39 10 0.7 1.75 9.52 0.4534 44.89 10 0.8 2.0 12.53 0.2404 23.80 -
[1] 郑浩, 彭毅, 王仕伟, 等. V2O5/石墨烯纳米复合材料的合成及储钠性能研究[J]. 钢铁钒钛, 2023, 44(1): 32-37.Zheng Hao, Peng Yi, Wang Shiwei, et al. Synthesis and sodium storage properties of V2O5/graphene nanocomposites[J]. Iron Steel Vanadium Titanium, 2023, 44(1): 32-37. [2] Huang Yangyang, Zheng Yuheng, Li Xiang, et al. Electrode materials of sodium-ion batteries toward practical application[J]. ACS Energy Letters, 2018,7(3):1604−1612. [3] Ni Qiao, Bai Ying, Wu Feng, et al. Polyanion-type electrode materials for sodium-ion batteries[J]. Advanced Science, 2017,24(1):90−114. [4] 周华, 宋永昌, 刘进, 等. 钒基电极材料在储能领域的研究进展[J]. 钢铁钒钛, 2022, 43(2): 73-80.Zhou Hua, Song Yongchang, Liu Jin, et al. Progress of vanadium-based electrode materials in energy storage[J]. Iron Steel Vanadium Titanium, 2022, 43(2): 73-80. [5] 张东彬, 常智, 滕艾均, 等. VN基材料的电子结构调控和超电容性能研究[J]. 钢铁钒钛, 2022, 43(5): 45-51.Zhang Dongbin, Chang Zhi, Teng Aijun, et al. Regulation on electronic structure of VN-based materials for enhanced supercapacitor performances[J]. Iron Steel Vanadium Titanium, 2022, 43(5): 45-51. [6] Chen Gongxuan, Huang Qing, Wu Tian, et al. Polyanion sodium vanadium phosphate for next generation of sodium-ion batteries—a review[J]. Advanced Functional Materials, 2020,30:2001289. doi: 10.1002/adfm.202001289 [7] Li Shuo, Dong Yifan, Xu Lin, et al. Effect of carbon matrix dimensions on the electrochemical properties of Na3V2(PO4)3 nanograins for highperformance symmetric sodium-ion batteries[J]. Advanced Materials, 2014,26:3545−3553. doi: 10.1002/adma.201305522 [8] Hao Xiaogang, Liu Zigeng, Gong Zhengliang, et al. In situ XRD and solid state NMR characterization of Na3V2(PO4)2F3 as cathode material for lithium-ion batteries[J]. Scientia Sinica Chimica, 2012,42(1):38−46. (郝小罡, 刘子庚, 龚正良, 等. 锂离子电池正极材料Na3V2(PO4)2F3的原位XRD及固体核磁共振研究[J]. 中国科学:化学, 2012,42(1):38−46. doi: 10.1360/032011-177Hao Xiaogang, Liu Zigeng, Gong Zhengliang, et al. In situ XRD and solid state NMR characterization of Na3V2(PO4)2F3 as cathode material for lithium-ion batteries[J]. Scientia Sinica Chimica, 2012, 42(1): 38-46 doi: 10.1360/032011-177 [9] Qi Yuruo, Tong Zizheng, Zhao Junmei, et al. Scalable room-temperature synthesis of multi-shelled Na3(VOPO4)2F microsphere cathodes[J]. Joule, 2018,2:2348−2363. doi: 10.1016/j.joule.2018.07.027 [10] Jiang Yu, Zhou Xuefeng, Li Dongjun, et al. Highly reversible Na storage in Na3V2(PO4)3 by optimizing nanostructure and rational surface engineering[J]. Advanced Energy Materials, 2018,8(16):1800068. doi: 10.1002/aenm.201800068 [11] Inoishi Atsushi, Setoguchi Naoko, Okada Shigeto, et al. Preparation of a single-phase all-solid-state battery via the crystallization of amorphous sodium vanadium phosphate[J]. Physical Chemistry Chemical Physics, 2022,24:27375−27379. doi: 10.1039/D2CP04328A [12] Peng Lele, Zhu Yue, Peng Xu, et al. Effective interlayer engineering of two-dimensional VOPO4 nanosheets via controlled organic intercalation for improving alkali ion storage[J]. Nano Letter, 2017,17:6273−6279. doi: 10.1021/acs.nanolett.7b02958 [13] Fang Yongjin, Yu Xinyao, Lou Xiongwen(David). Nanostructured electrode materials for advanced sodium-ion batteries[J]. Matter, 2019,1(1):90−114. doi: 10.1016/j.matt.2019.05.007 -





 下载:
下载: